Hess’s Law of constant heat summation
It may be stated as follows:
If a chemical transformation can occur through several pathways, whether in a single step or in several stages, the total heat change remains constant regardless of the technique employed to effectuate the change.
Hess’s Law can feel like a maze of math, but at its heart, it’s just the “Law of Conservation of Energy” applied to chemistry. It tells us that the total energy change in a reaction is like a mountain hike: whether you take the steep direct path or the winding scenic trail, the change in altitude from start to finish is exactly the same.
In chemistry, we call this a “State Function.” It means the route doesn’t matter; only the starting reactants and the final products do.
The General Law: Multiple Routes, One Destination
Hess’s Law can feel like a maze of math, but at its heart, it’s just the Law of Conservation of Energy applied to chemistry. It tells us that the total energy change in a reaction is like a mountain hike: whether you take the steep direct path or the winding scenic trail, the change in altitude from start to finish is exactly the same.
In chemistry, we call this a “State Function.” This means the enthalpy change is independent of the pathway taken; it only depends on the initial state of the reactants and the final state of the products.
The General Law: Multiple Routes, One Destination
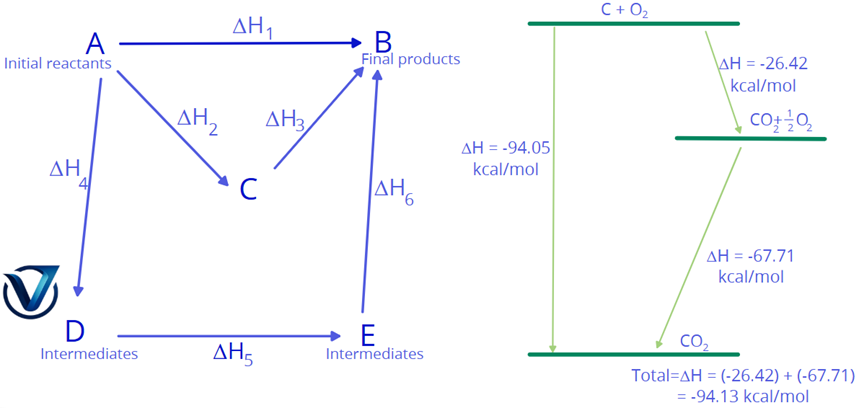
Imagine you want to transform Substance A into Substance B. According to Hess’s Law, the enthalpy change (ΔH) will be identical regardless of the complexity of the reaction path.
- Route 1 (Direct): A → B (Change = ΔH1)
- Route 2 (Two-Step): A → C → B (Change = ΔH2 + ΔH3)
- Route 3 (Multi-Step): A → D → E → B (Change = ΔH4 + ΔH5 + ΔH6)
ΔHDirect = ∑ ΔHSteps
Practical Example: Carbon Dioxide (CO2) Formation
To see this in action, we can look at the formation of Carbon Dioxide. It can be produced in one single step or through a two-stage process involving Carbon Monoxide.
1. The Direct Route:
Carbon reacts with oxygen to form CO2 immediately:
C(s) + O2(g) → CO2(g) ΔH = -393.5 kJ/mol
2. The Indirect Route:
First, Carbon Monoxide is created, which is then further oxidized:
The Result: By adding the steps together (-110.5 kJ + -283.0 kJ), we get -393.5 kJ. This perfectly matches the direct route, proving that energy remains constant regardless of the path.
Born-Haber cycle
The Born-Haber cycle is a thermochemical cycle used primarily to calculate the lattice energy of ionic compounds, which is otherwise difficult to measure directly. Named after Max Born and Fritz Haber, it applies Hess’s Law to the formation of an ionic solid from its constituent elements. The cycle treats the overall enthalpy of formation as a multi-step process involving several distinct energy changes, including enthalpy of atomization, ionization energy, electron affinity, and sublimation. By visualizing these steps in a closed loop, scientists can determine the “missing” energy value—usually the lattice enthalpy—by ensuring the total energy change across the alternative pathways equals the standard enthalpy of formation.
To calculate the lattice energy, we sum the energy required for each stage:
Variable Definitions
- ΔHf° : The standard enthalpy of formation.
- ΔHat° & ΔHsub° : Represent atomization and sublimation energies.
- IE & EA : Ionization Energy and Electron Affinity respectively.
- ΔHlattice : The lattice enthalpy you are typically solving for.
Born Haber Cycle for NaCl
Enthalpy changes for direct synthesis
The enthalpy change for the direct synthesis of NaCl from sodium metal and chlorine gas is -114 kJ.
Na(s) + Cl2 NaCl(s) ΔH° = -114 kJ
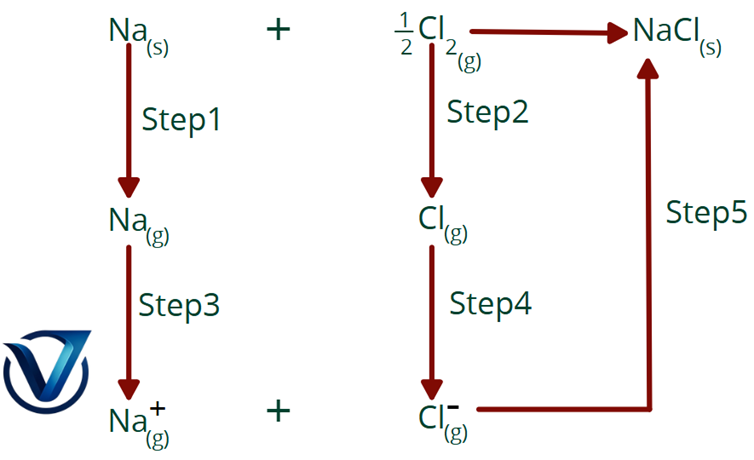
Enthalpy alterations using indirect methods
The direct enthalpy change of NaCl mentioned above is equivalent to the enthalpy change of the five processes involved in the production of NaCl.
Step 1. Sublimation of sodium metal to gaseous atoms.
Na(s) Na(g) ΔH°1 = +108kJ
Step 2. Dissociation of chlorine molecules into chlorine atoms. The enthalpy of dissociation is 121 kJ/mol for chlorine.
Cl2(g) Cl (g) ΔΗ°2 =+121 kJ
Step 3. Conversion of gaseous sodium to sodium ions by electron loss. The enthalpy of ionisation, or ionisation energy, is 495 kJ per mole of sodium atoms.
Na(g) Na+(g) + e– ΔΗ°3 =+495kJ
Step 4. Chlorine atoms get an electron to generate chloride ions. The energy released corresponds to the electron affinity of chlorine, quantified as
Cl + e– Cl–(g) ΔΗ°4 =- 348kJ
Step 5. Sodium and chloride ions combine to create a crystal lattice. The energy in this operation is the inverse of lattice energy.
Na+(g) + Cl– (g) → NaCl(s) ΔΗ°5 = – (lattice energy)
The lattice energy may be determined by equating the enthalpy of creation of NaCl, -411 kJ, to the cumulative enthalpy changes of the five phases.
ΔΗ°1 + ΔΗ°2 +ΔΗ°3 + ΔΗ°4 + ΔΗ°5 =- 411kJ
108 kJ + 121 kJ + 495 kJ – 348 kJ – (lattice energy) = -411 kJ
Solving this equation yields
Lattice energy: +787 kJ/mol
The sequence of transformations seen in the aforementioned figure is referred to as the Born-Haber Cycle, named after Max Born and Fritz Haber, who developed this technique for estimating lattice energies.
Watch Complete Lecture in Urdu/Hindi for comprehensive understanding
Download Complete Notes Below
Proudly Powered By




Leave a Comment