Debye-Hückel Theory Visualizer
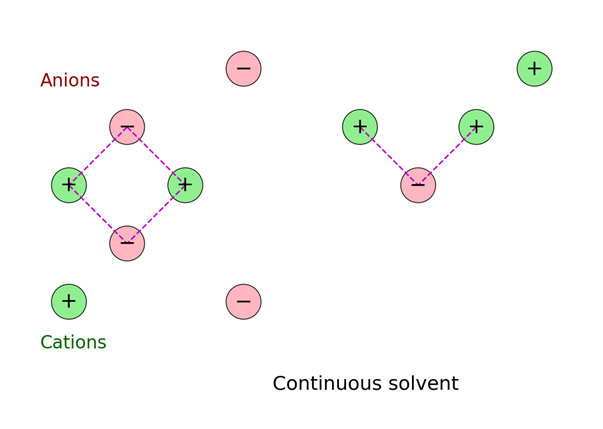
This ionic atmosphere screens the central ion’s charge, causing the electrostatic potential to decrease exponentially with distance, characterized by the Debye length (λD).
Blue dots = counter-ions (negative charge, attracted), red dots = co-ions (positive charge, repelled), and the large dark blue dot is the central ion.
Postulates of Debye-Hückel-Onsager Theory
The Debye-Hückel-Onsager (DHO) theory refines our understanding of how strong electrolytes behave in solution, moving beyond the simple Arrhenius model.
The Debye-Hückel-Onsager Equation
For a completely dissociated univalent electrolyte, the relation between conductance and concentration is expressed as:
Understanding the Constants A and B
These constants account for the physical forces that "drag" or slow down the ions in solution:
Measures the Electrophoretic Effect.
(For water at 25°C, A = 60.20).
Measures the Asymmetry Effect (Relaxation Effect).
These constants depend on the dielectric constant (D) and the coefficient of viscosity (η) of the medium at absolute temperature.
Watch Full lecture in Urdu/Hindi
Watch Full lecture in English
The Debye-Hückel Theory provides the theoretical foundation for explaining why electrolyte solutions deviate from ideal behavior. This deviation is primarily caused by interionic electrostatic forces. In dilute solutions, these forces create a surrounding "ionic atmosphere" around each individual ion, which effectively lowers its chemical potential. This interaction results in an activity coefficient (γ) that is less than one.
Activity Coefficients and the Limiting Law
Because solutions must maintain electroneutrality, individual ion activities cannot be monitored directly. Instead, the theory focuses on determining the mean ionic activity coefficient (γ±).
Where:
- z is the ionic charge.
- I is the ionic strength of the solution.
- A is a constant (approximately 0.509 for water at 25°C).
Divergence from Ideal Behavior: Case Comparisons
Even at identical concentrations, different electrolytes show varying degrees of non-ideal behavior based on their ionic charges.
| Electrolyte | Concentration | Ionic Charge (z) | Activity Coefficient (γ±) |
|---|---|---|---|
| NaCl | 0.01 M | +1, -1 | ≈ 0.90 |
| MgCl2 | 0.01 M | +2, -1 | ≈ 0.72 |
Note: The magnesium ion’s higher charge (+2) results in a far greater divergence from optimal behavior compared to the sodium ion (+1), even when molar concentrations are the same.
Step-by-Step Calculation for 0.01M NaCl
To calculate the activity coefficient, we must first determine the ionic strength of the solution.
Extended Debye-Hückel Theory
When dealing with higher concentrations where finite ion size (the distance of closest approach, α) becomes significant, the extended form is employed:
This extended theory typically applies to ionic concentrations up to approximately 0.1 mol/kg.
Download Complete Notes Below
Proudly Powered By




Leave a Comment