Kohlrausch’s law states that at infinite dilution, each ion contributes a fixed amount to the molar conductivity, independent of other ions. The total conductivity of an electrolyte is the sum of the contributions of its cations and anions. It is used to determine limiting molar conductivity, ion mobilities, and dissociation constants.
At infinite dilution, the equivalent conductance of an electrolyte is equal to the sum of the equivalent conductances of its individual ions.
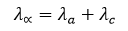
Here, the equivalent conductance of the anion and the cation are denoted separately.
For example, at 25°C, the equivalent conductance of NaCl at infinite dilution is found to be 126.45 ohm⁻¹ cm² eq⁻¹.
The equivalent conductance of the Na⁺ ion is 50.11 ohm⁻¹ cm² eq⁻¹, and that of the Cl⁻ ion is 76.34 ohm⁻¹ cm² eq⁻¹.
Watch full lecture in Hindi/Urdu
Watch full lecture in English
Applications of Kohlrausch’s Law
(1) Calculation of λ∞ for Weak electrolytes
Weak electrolytes do not ionize significantly in solution and remain largely unionized even at very high dilution. As a result, it is not practically possible to determine their equivalent conductance at infinite dilution (Λ∞) through direct measurement. However, Λ∞ can be calculated indirectly using Kohlrausch’s Law of Independent Migration of Ions.
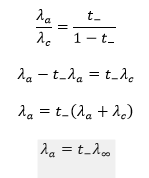
The ionic conductance of an ion can be determined by multiplying the equivalent conductance at infinite dilution of a strong electrolyte containing that ion by its transport number.
Using this method, the ionic mobilities of both ions in a weak electrolyte can be calculated.
By adding the conductances of the individual ions, the equivalent conductance at infinite dilution (Λ∞) of the weak electrolyte can then be obtained.
(2) Calculation of Absolute Ionic mobilities
The absolute ionic mobility of an ion is defined as the velocity of an ion in centimetres per
second under a potential gradient of one volt per centimetre. (Potential gradient = applied EMF/
distance between (the electrodes). It is expressed in cm sec–1.
For example, if the velocity of an ion at infinite dilution is cm/s, and the distance between the electrodes is 20 cm with an applied voltage of 100 V, the potential gradient is 100/20 i.e., 5 volts per cm. Therefore, the ionic mobility is U/5 cm sec–1.
(3) Calculation of the solubility of sparingly soluble salts
Determination of the solubility of sparingly soluble salts
AgCl and PbSO4, two substances that are typically said to as insoluble, have a specific water solubility value. Conductance measurements of their saturated solutions can be used to ascertain this. Even in a saturated solution, the extremely little amount of solute must entirely dissolve into ions in order for the equivalent conductance κV to equal the equivalent conductance at infinite dilution. Kohlrausch’s Law states that this is the total of the ionic mobilities.
κV = λ ∞ = λ anion + λ cation
It is possible to determine V, which is the volume in millilitres holding one gram-equivalent of the electrolytes, by knowing κ and λ∞.
Download Complete Notes for Kohlrausch’s Law Below
(4) Calculation of the Degree of Dissociation or Conductance Ratio
An electrolyte’s apparent degree of dissociation, α, at dilution V can be found using the formula, α * λv/λ∞ where λv is the electrolyte’s equivalent conductance at dilution V, and λ∞ is its equivalent conductance at infinite dilution.
Kohlrausch’s Law states that this is equal to the sum λ anion and λ cation.
(5) Calculation of the Ionic product for water
The observed specific conductance of the purest water at 25oC is 5.54 × 10-8 mho cm-1 . The conductance of one litre of water containing 1 gram eqvt of it would be:
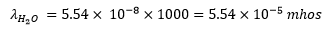
When the temperature is the same, the conductance of H+ ions andOH– ions is:
λH+ = 349.8 mho cm-1
λ OH– = 198.5 mho cm-1
According to Kohlrausch’s Law
λH2O = λH+ + λ OH–
349.8 + 198.5 = 548.3 mho cm-1
Since one water molecule gives one H+ ion and one OH– ion
So
H2O = H+ + OH –
Assuming that conductance and ionic concentration are proportionate, we have
[H+] = [OH–] = (is 5.54 × 10-8) / 548.3= 1.01 × 10-7 g ion litre-1
The ionic product of water is then
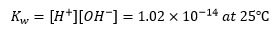
For most purposes, the value of Kw is taken to be 10-14
Proudly Powered By




Leave a Comment