Electrolytic Conductance: Ohm’s Law and Resistance Factors in Solutions
In electrochemistry, Conductance (or conductivity) is defined as “the ability of electrolytes to allow electric current to pass through them.” Unlike metallic conductors where electrons flow, in electrolytes, the current is carried by ions.
1. Ohm’s Law in Electrolytic Solutions
Interestingly, electrolytes follow Ohm’s Law just like metals. The electric current (I) moving through an electrolyte is directly proportional to the potential difference (E or V) applied across the electrodes.
- I: Current (measured in Amperes)
- E: Potential difference across the electrodes (Voltage)
- R: Resistance (measured in Ohms, Ω)
Note: Electrolytes behave ohmically only when concentration and temperature are maintained, and the applied voltage is kept low.
2. Factors Determining Resistance (R)
The resistance of an electrolyte solution is not fixed; it depends on several geometric and material physical conditions:
Mathematical Relationship:
R = ρ (l / A)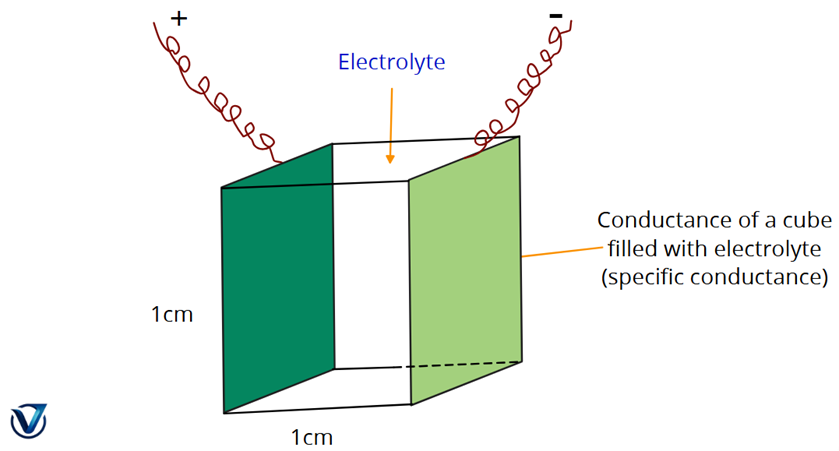
Understanding Resistivity and Specific Conductivity
Different electrolytes naturally resist the flow of electricity to varying degrees. This inherent property is called Resistivity (ρ). While some act as good conductors with low resistivity, others act as insulators with high resistivity.
🚗 The Everyday Analogy
Think of electricity as cars on a road:
- 🛣️ Longer Road (l): Harder to travel (More Resistance).
- 🛤️ Wider Road (A): Easier to travel (Less Resistance).
- 🚧 Road Surface (ρ): Smooth asphalt (Low Resistivity) vs. Muddy track (High Resistivity).
Specific Conductance (κ)
The reciprocal (inverse) of resistivity is called Specific Conductivity. It measures how easily current flows through a specific material.
💡 Student Challenge: Logic Test
Question: If the distance between two electrodes in an electrolyte solution is doubled, but their surface area remains the same, what happens to the resistance?
Analysis:
- In a solution, length (l) is the distance between electrodes.
- Area (A) is the surface area in contact with the solution.
- Resistance is directly proportional to length (R ∝ l).
The Verdict: Doubling the distance between electrodes doubles the resistance. Since the electrode area remains constant, it provides no counter-balancing change.
Watch full lecture in Hindi/Urdu
Watch full lecture in English
Download Complete Notes Below
Measurement of Electrolytic conductance
Conductance is the reciprocal of resistance, meaning it can be calculated by measuring the resistance of an electrolytic solution. This measurement is typically carried out in the laboratory using a Wheatstone bridge. The solution whose conductance is to be measured is placed in a specialized container called a conductance cell. A basic type of conductance cell commonly used in laboratories is illustrated in Fig. 24.6. The cell contains platinum electrodes coated with platinum black, which are attached to platinum wires sealed within two narrow glass tubes. Electrical contact with the circuit’s copper wires is established by immersing them in mercury contained within these tubes. Electrical contact with the copper wires of the circuit is established by immersing them in mercury contained within the glass tubes.

The setup commonly used to measure the resistance of a conductance cell is illustrated in Fig. 24.7. In this arrangement, a headphone is used instead of a galvanometer to detect the balance point. AB represents a manganin wire that is tightly stretched along a meter scale marked in millimeters. A sliding contact, denoted by H (indicated with an arrow), can move along the wire. The component labeled R is a resistance box, while C is the conductance cell containing the electrolytic solution. An induction coil, labeled I, supplies alternating current to the circuit as shown in the diagram.

When current flows, all resistances in the resistance box are disconnected. The sliding contact is then adjusted until the sound in the headphones is at a minimum. At this point, the system is in balance, and we have:

The resistance of a solution measured using the conductance cell can be converted to specific conductance using the equation:

The ratio l/A has been put equal to x. That is,

The value of is constant for a given cell and is referred to as the cell constant. Once the specific conductance ( ) is determined, the equivalent conductance (Λ) and the molar conductance of the solution can be calculated using the relevant formulas.

Where N is the gram-equivalent and M is the gram-mole of the electrolyte.
Determination of the Cell constant
The exact value of the cell constant ( ) can be determined by measuring the distance between the electrodes ( ) and their cross-sectional area ( ). However, accurately measuring these dimensions is quite difficult. Therefore, an indirect method is commonly used to determine the cell constant.
We know that:

To determine the cell constant, a standard solution of potassium chloride (KCl) with a known specific conductance at a given temperature is used. A KCl solution of the same concentration is then prepared, and its conductance is measured experimentally at the same temperature.
Proudly Powered By




Leave a Comment