Conductometric titrations are those in which conductance measurements are used to determine the end-point of acid-base reactions, certain displacement reactions, or precipitation reactions.
The conductometric titration is a quantitative analytical technique that estimates the concentration of an analyte by measuring changes in a solution’s electrical conductivity as a titrant is added. The endpoint is shown by a visible change in conductivity when high-mobility ions are replaced by lower-mobility ones, or vice versa.
These titrations rely on the fact that the conductance of a solution at a constant temperature depends on the number of ions present and their mobility.
Theory of Conductometric Titrations
The theory behind conductometric titrations states that the end-point can be determined through conductivity measurements. In an acid-base neutralization, the initial addition of the base decreases the solution’s conductivity, as highly conductive H⁺ ions are replaced by the less conductive cations of the base.
After the equivalence point is reached, the concentration of ionic species increases, leading to a rise in the solution’s conductance. When conductance values are plotted against the volume of titrant added, two straight lines with opposite slopes are obtained. The point of intersection of these lines represents the equivalence point.
Principle of Conductometric Titrations
During a titration, one ion is replaced by another, and the difference in their ionic conductivities directly affects the overall conductivity of the solution.
It can also be observed that ionic conductance values differ between cations and anions. Ultimately, the conductivity of the solution also depends on whether a chemical reaction occurs within the electrolyte.
Essential Concepts of Conductometric Titrations
- Ion Mobility: Conductivity is determined by the amount and mobility of ions present. Because of their high mobility, H+ and OH- ions have higher conductivity than other ions.
- Equivalency Point: Conductive ions are either consumed or replaced by other ions as the titrant is added. The equivalence point on a volume added vs. conductivity graph is known as the inflection point.
- Mechanism: For instance, conductivity initially decreases when Na+ (slow) replaces H+ (fast) in a titration of HCl (strong acid) vs NaOH (strong base). After the endpoint, the conductivity increases due to an excess of OH-ions.
Examples of conductometric titration
Strong base against strong acid: NaOH versus HCl
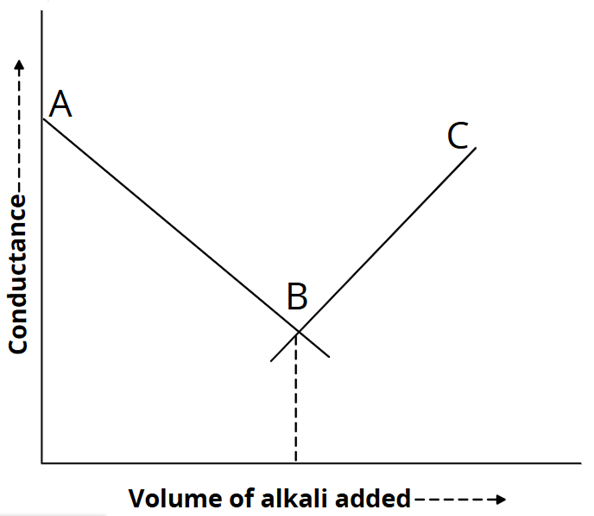
Acid is put in a conductivity beaker, and alkali in a burette. The conductance of HCl is caused by the ions H+ and Cl−. As alkali is provided, slow-moving Na+ ions gradually take the place of H+ ions, as illustrated below:
H+ + Cl– + Na+ + OH– → Na+ + Cl– + H2O
Conductance decreases with the addition of NaOH until complete neutralisation. If alkali is supplied after the end point, fast-moving OH– ions will be introduced. Consequently, the conductivity increases when more alkali is introduced. Two straight lines that intersect at point “B,” the titration’s end point, are produced when the conductance variation is plotted against the volume of alkali added.
Watch Full lecture in Urdu/Hindi
Watch Full lecture in English
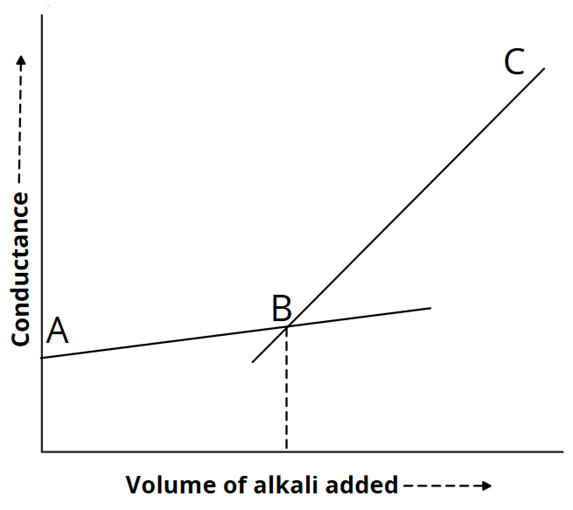
2) CH3COOH versus NaOH: weak acid versus strong base
The conductance steadily increases up to the equivalence point when a strong base is introduced to a weak acid, changing the poorly conducting acid into a highly ionised salt. The addition of alkali causes a notable rise in conductance above the equivalence point because of an excess of hydroxide ions.
CH3COO– + H+ + Na+ + OH– → CH3COO– + Na+ + H2O
The graph appears as:
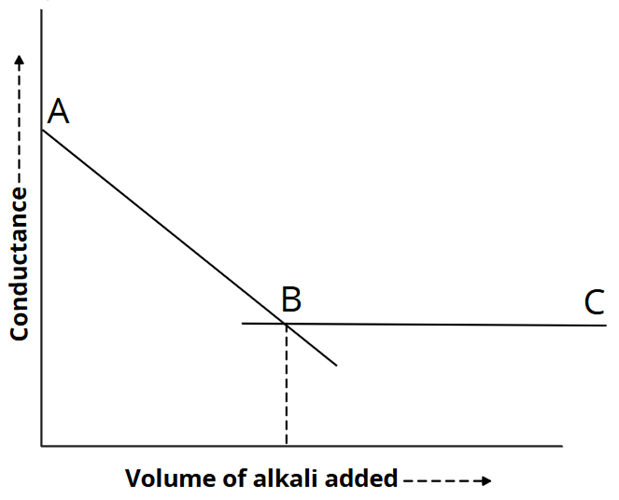
3) Strong acid vs weak base: NH4OH versus HCl
In this case, conductance first decreases when fast-moving H+ ions are replaced with slow-moving NH4+ ions.
H+ + Cl– + NH4+ + OH– → → NH4+ + Cl– + H2O
Beyond the endpoint, conductance will not be considerably changed by weaker ionised NH4OH. The intersection of the curves marks the end of the titration.
4) Weak base vs weak acid: CH3COOH versus NH4OH
In this titration, conductance first increases because salt (CH3COONH4), a strong electrolyte, is produced. Until the end, this increment continues. After the termination point, the conductance does not change noticeably.
H+ + NH4+ + OH– → → CH3COO– + NH4+ + H2O
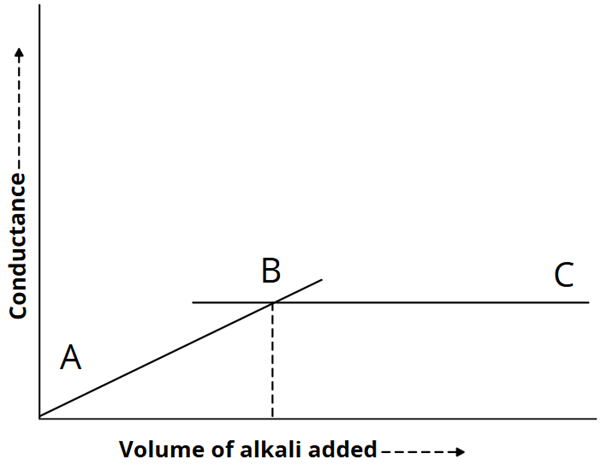
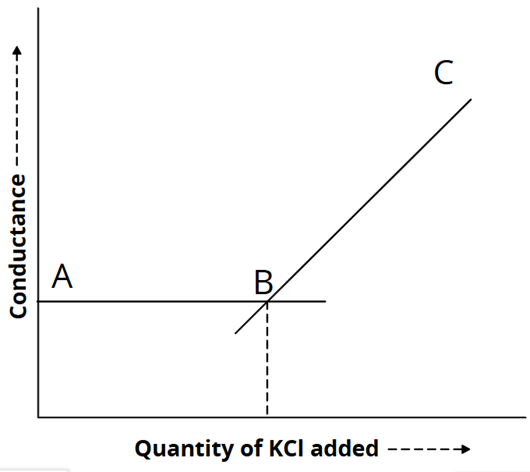
5) Precipitation titration: (AgNO3 against KCl)
The titration of silver nitrate against potassium chloride involves precipitate formation.
AgNO3 + KCl → KNO3 + AgCl
Since the ionic mobilities of Ag⁺ and K⁺ are nearly the same, the conductance remains almost constant up to the equivalence point. Beyond this point, the addition of KCl causes a rapid increase in conductance, as illustrated in the graph below.
Advantages of conductometric titrations:
1. Only a small quantity of solution is required for conductometric titrations.
2. Since the endpoint is determined graphically, no special precautions are needed.
3. Indicators are not required, making these titrations suitable for coloured or turbid solutions.
4. Conductometric titrations are effective for analyzing dilute solutions and weak acids.
5. They can be applied to mixtures of acids, precipitation reactions, and various other types of titrations.
6. Conductometric measurements offer higher accuracy in results.
Disadvantages of conductometric titrations:
1. High concentrations of salt in the solution can affect accuracy and lead to unreliable results.
2. The presence of other electrolytes besides the target analyte can interfere with the measurements and reduce accuracy.
3. Conductometric titrations have limited applicability in redox reactions.
Applications of conductometric titrations:
Applications of Conductometric Titration:
1. Detection of water pollution.
2. Quantitative analysis of various chemical compounds.
3. Determination of water alkalinity.
4. Measurement of water salinity.
5. Evaluation of the basicity of organic acids.
6. Estimation of deuterium ion concentration in water.
7. Determination of the solubility of sparingly soluble salts.
Download Complete Notes Below
Proudly Powered By




Leave a Comment