What is Heat Capacity?
Heat capacity or thermal capacity is a broad feature of matter that delineates its physical characteristics.
The heat required to raise the temperature of a certain mass of a substance by one unit without inducing a phase transition is termed heat capacity (C) or thermal capacity. It delineates a substance’s capacity to absorb thermal energy.
Heat capacity for a unit mass of a material is mathematically described as,
C = ΔQ / ΔT
where ΔQ represents the quantity of heat delivered to the substance and ΔT denotes the change in temperature.
Specific Heat Capacity
Specific heat capacity is defined as the number of heat changes i.e., heat received or rejected by a substance per unit mass in order to change its temperature by one unit.
If C represents the heat absorbed or rejected (ΔQ) by a substance of mass m during a temperature change ΔT, the specific heat capacity of that substance is defined as
S = C/m = (1/m) (ΔQ/ΔT).
Molar Heat Capacity
The quantity of heat required to raise the temperature of one mole of any material by one degree Kelvin is termed the Molar Heat Capacity. Molar heat capacity and specific heat capacity are nearly identical, with the primary exception being that specific heat capacity is quantified per gram, whereas molar heat capacity is quantified per mole.
Units: Molar Heat Capacity is quantified in Joules per mole.
The equation for calculating molar heat capacity is,
Q = n C∆T
where Q represents the heat provided, n denotes the moles of the substance, and C signifies the molar heat capacity of the material.
Molar Heat Capacity at Constant Pressure, Cp
The heat necessary to increase the temperature of one mole of a substance by one degree Kelvin at constant pressure is referred to as the Molar Heat Capacity at Constant Pressure, written as Cp.
The first law of thermodynamics states that
dq = dE + PdV.
By dividing both sides by dT, we obtain
dq/dT = dE/dT + PdV/dT
We know H = E + PV
By differentiating this equation with respect to T at constant pressure, we obtain
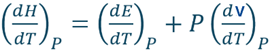
Comparing it with, dq/dT = dE/dT + PdV/dT we get
Consequently, heat capacity at constant pressure is defined as the derivative of enthalpy with respect to temperature at constant pressure.
Molar Heat Capacity at Constant Volume (Cv)
The heat necessary to increase the temperature of one mole of any substance by one degree Kelvin at constant volume is referred to as the Molar Heat Capacity at Constant Volume, written as Cv.
According to the first rule of thermodynamics,
dq = dE + PdV.
By dividing both sides by dT, we obtain
dq/dT = (dE + PdV)/dT
At constant volume, where dV = 0, the equation simplifies to
Cv = (dE/dT) v.
The heat capacity at constant volume is defined as the derivative of internal energy with respect to temperature at constant volume.
Ratio of Heat Capacity
The ratio of heat capacity at constant pressure (Cp) to heat capacity at constant volume (Cv) is referred to as the Heat Capacity Ratio.
The Greek sign γ denotes it.
γ = Cp / Cv
Correlation Between Cp and Cv
According to the first rule of thermodynamics:
ΔQ = ΔU + ΔW
where ΔQ represents the heat transferred to the system, ΔU denotes the change in internal energy, and ΔW signifies the work performed.
We may express
ΔQ as ΔU + PΔV,
which can be rewritten as
ΔW = PΔV
Given that
ΔQ = nCpΔT and ΔU = nCvΔT.
Consequently
nCpΔT = nCvΔT + PΔV
Given that
PV = nRT
At T1 Kelvin:
PV1 = nRT1 ———— (a)
At T2 Kelvin:
PV2 = nRT2 ———— (b)
Subtracting equation (a) from (b):
PV2 – PV1 = nRT2 – nRT1
P (V2 – V1) = nR (T2 – T1)
Here,
V2 – V1 = ΔV and T2 – T1 = ΔT
Therefore
PΔV=nRΔT
By substituting the value of PΔV into equation (3), we obtain:
nCpΔT = nCvΔT + nRΔT
which simplifies to
nCpΔT = nΔT (Cv + R).
Cp = Cv + R
Alternatively,
Cp − Cv = R
The subsequent connection may be established by studying the ideal gas behaviour of a gas.
Cp – Cv = R
Here, R denotes the universal gas constant.
What is the reason for Cp being more than Cv?
Cp and Cv represent the specific temperatures of an ideal gas. These denote the amount of heat required to elevate the temperature of a unit mass by 1°C.
By the first rule of thermodynamics
ΔQ=ΔU+ΔW
Where, ΔQ represents the quantity of heat supplied to the system, ΔU denotes the variation in internal energy, and W signifies the work done.
So, with constant pressure, heat is absorbed not just for increasing the internal energy (function of temperature) but for producing work as well. At constant volume, heat is absorbed solely to increase internal energy and not to do any work on the system, as is the case for a closed system.
W = PΔV
where W represents the labour performed. In this context, ΔV=0 (A closed system is a fundamental prerequisite for maintaining constant volume). Consequently, the specific heat at constant pressure exceeds that at constant volume, i.e.,
Cp > Cv
Watch Complete Lecture in Urdu/Hindi for comprehensive understanding
Download Complete Notes Below
Proudly Powered By




Leave a Comment