Equilibrium Constant & Entropy & Free Energy
Understanding the spontaneity of a chemical system requires looking at the universe as a whole. The total entropy change is the ultimate decider of whether a reaction will proceed.
Recall that ΔSᶿtotal must be positive for all spontaneous processes.
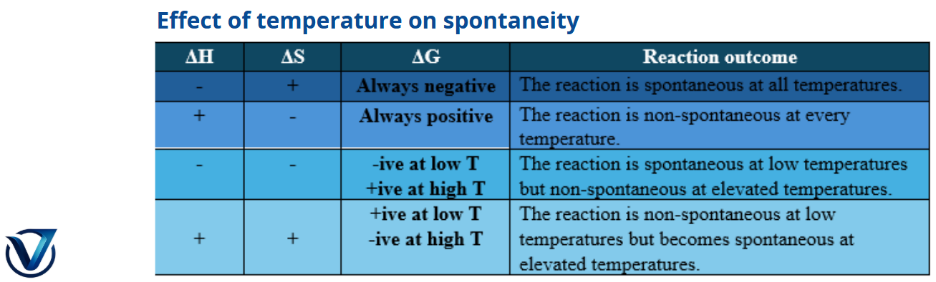
• System Entropy (ΔSᶿsys): Exhibits minimal variation with temperature alterations unless a phase transition occurs in the reactants or products.
• Surroundings Entropy (ΔSᶿsurr): A substantial alteration is anticipated based on heat exchange with the environment.
Quantifying the Surroundings
The entropy variation of the surroundings during a chemical process is directly related to the enthalpy change and absolute temperature:
Where ΔH represents the enthalpy change and T denotes the absolute temperature (measured in kelvins).
This information is essential for chemists to ascertain the spontaneity of a reaction at any specific temperature.
The Equilibrium Relationship
Consider a system reaching a state of balance. For instance, examine the subsequent reaction:
N₂O₄ (g) ⇌ 2NO₂ (g)In a reversible reaction capable of attaining equilibrium, the equilibrium position can be approached from either direction. This leads to a fascinating thermodynamic reality:
- Both the forward and reverse responses are spontaneous as the system moves toward equilibrium.
- Consequently, ΔS must be positive in both orientations during the approach to equilibrium.
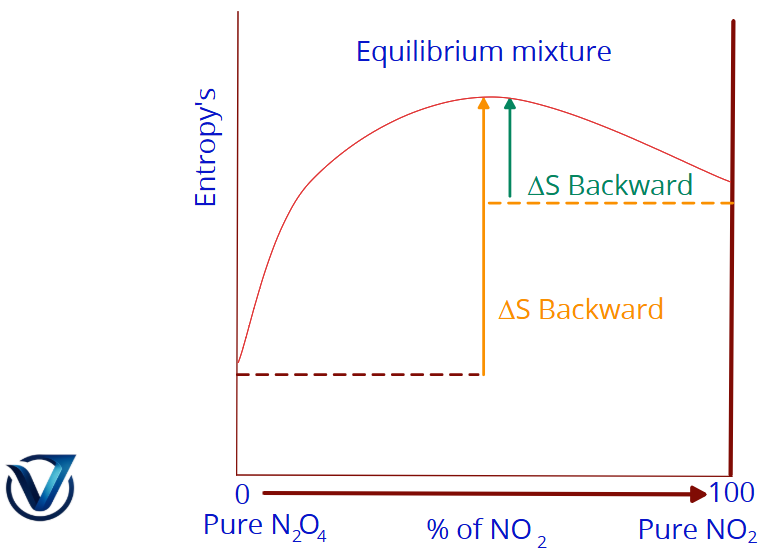
Entropy Dynamics in the N₂O₄ ⇌ 2NO₂ System
The behavior of entropy in a chemical mixture is a critical indicator of how a system reaches balance. Consider the relationship between entropy and the composition of a mixture of N₂O₄ and NO₂:
Graph: Entropy (S) reaching its maximum at the equilibrium position.
- The entropy of N₂O₄ is inferior to that of the equilibrium mixture.
- The entropy change from pure N₂O₄ to the equilibrium mixture is positive; therefore, the change is spontaneous.
- The entropy change during the transition of NO₂ to the equilibrium mixture is also positive, making this alteration likewise spontaneous.
Regardless of the starting proportions, the entropy change for a gas mixture is positive as it approaches equilibrium. At equilibrium, the cumulative entropy change is zero, meaning:
Neither the forward nor the reverse reaction can reach completion because the entropy shift from the equilibrium mixture back to pure reactants or pure products is negative.
Entropy & The Equilibrium Constant
The correlation between the total entropy of the reaction and the equilibrium constant (Kc or Kp) is mathematically expressed as:
To calculate the equilibrium constant using the total entropy change, we rearrange the equation:
K = e(ΔStotal / R)
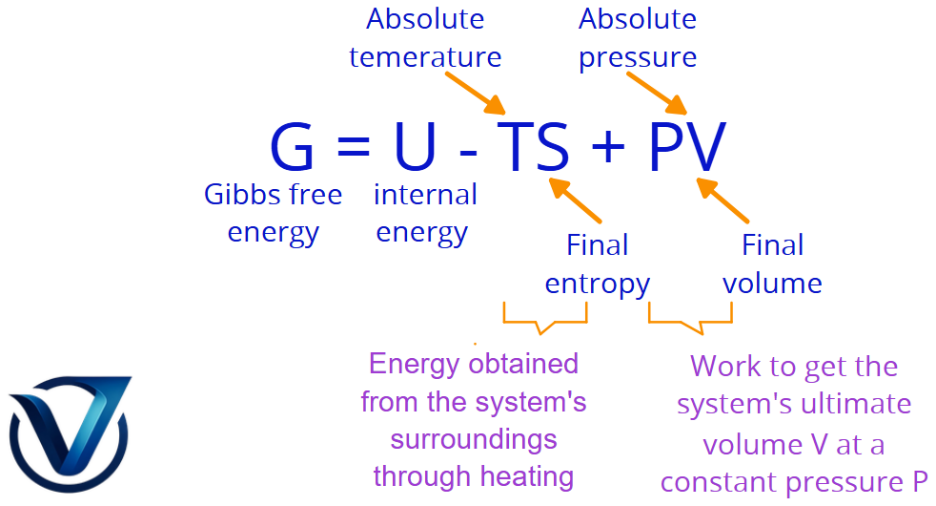
Gibbs Free Energy (G)
Gibbs free energy represents the maximum energy a material may provide for a chemical transformation or reaction. It acts as the aggregate of entropy and enthalpy within a closed system.
G = U + PV – TS
The change in energy, ΔG, signifies the direction of a chemical reaction under two specific conditions: constant pressure and constant temperature.
Standard Units & Definitions:
Correlation between ∆G and spontaneity
1. ∆G < 0 indicates a spontaneous process in the forward direction, characterized by the release of energy.
2. ∆G > 0 indicates a nonspontaneous process in the forward direction.
3. ∆G = 0 indicates that the system is in equilibrium; no net change transpires.
Equilibrium Constant
- The equilibrium constant is the ratio of product concentrations to reactant concentrations. It is represented by K(eq). The equation for the equilibrium constant is:
Here K(c) denotes the equilibrium constant expressed in moles per liter.
The connection between Gibbs free energy and the equilibrium constant
At equilibrium, there is no further change in free energy, i.e., ΔG = 0, and the reaction quotient (Q) equals the equilibrium constant (K(eq)). Thus, the correlation between K(eq) and ΔG is established as follows:
ΔG° = -RT ln K(eq) or ΔG° = -2.303 RT log K(eq)
- Where R denotes the ideal gas constant (8.314 J/K mol)
- T represents the temperature in Kelvin.
- K(eq) represents the natural logarithm of the equilibrium constant
Variations in Free Energy, AG, during a Reaction
1. A significant negative value of ΔG° (ΔG° < 0) indicates a spontaneous process, wherein the reaction mixture predominantly consists of products.
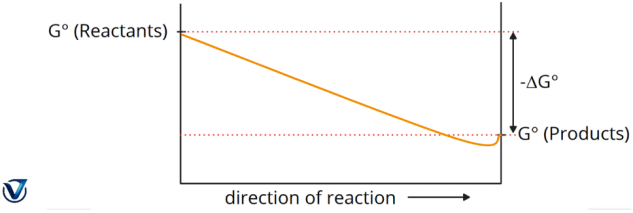
2. ΔG° is a substantial positive value (ΔG° > 0). Nonspontaneous process: the reaction mixture predominantly consists of reactants.
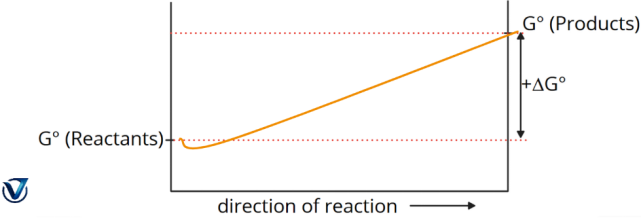
- At the minimum point, this represents the equilibrium mixture of reactants and products. G equals zero.
- The real quantity of free energy (ΔG) fluctuates as the reaction progresses, although the standard free energy change (ΔG°) remains constant.
Correlation between ΔG and ΔG°
ΔG = ΔG° + RT ln(Q)
At equilibrium, ΔG = 0 and Q = K:
ΔG° = -RTInK
on rearranging
Correlation between ΔG° and K
1. ΔG°is negative: K > 1 indicates that the products are favoured.
2. ΔG°equals zero: K equals 1.
3. ΔG° is positive: K < 1 (reactants favoured)
Test your learning 💡
Download Complete Notes Below
Proudly Powered By




Leave a Comment