A state of matter refers to one of the several forms of matter. Four states of matter are observable in daily life: solid, liquid, gas, and plasma. Numerous intermediate states, including liquid crystals, are recognized, but specific states, like as Bose-Einstein condensates, neutron-degenerate matter, and quark-gluon plasma, are acknowledged to occur solely under extreme circumstances, including extreme cold, excessive density, and extreme energy.
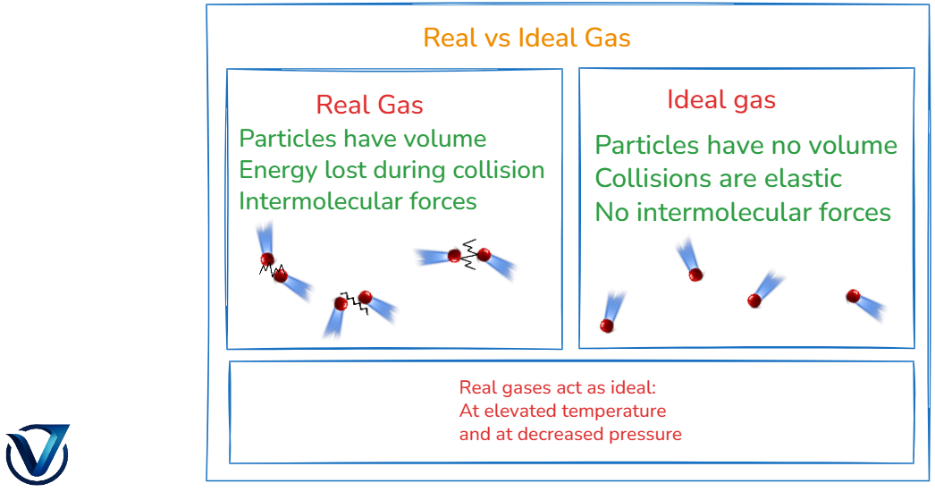
| Comparison between Real and Ideal gas | |
|---|---|
| Ideal Gas | Real Gas |
| Have no definite volume | Have definite volume |
| Show elastic collusion between particles | Show non-elastic collusion between particles |
| No intermolecular forces | Have intermolecular forces |
| These are hypothetical | Exist in the environment |
| Independent from exitance of other gases | Interact with each other in a mixture |
| They do not depend upon pressure and temperature | They become ideal at low pressure and high temperature |
| Obeys gas laws | Don’t obey gas laws |
Behavior of Real Gases
All gases are tangible examples. While a perfect gas does not exist, real gases are seen to exhibit ideal behavior under specific conditions. Nitrogen, oxygen, hydrogen, carbon dioxide, helium, and other genuine gases serve as examples.
Ideal Gas and Real Gas:
An ideal gas obeys the ideal gas equation PV=nRT under all pressures and temperatures. Conversely, no gas is exceptional. Nearly all gases deviate from optimal behavior in some respect. Non-ideal or real gases, including H2, N2, and CO2, do not conform to the ideal gas law.
Compressibility Factor:
A novel parameter known as the Compressibility factor, represented as Z, serves to measure the extent of deviation of actual gases from ideal behavior. It is expressed as
Z = PV/RT
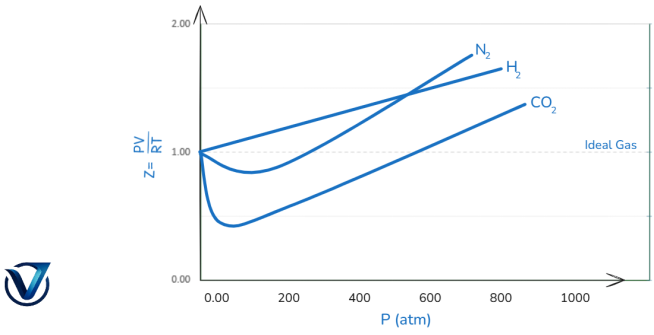
Graphing the compressibility factor, Z, against pressure, P, illustrates the extent of deviation from ideal behavior. Temperature and pressure do not influence an ideal gas, which possesses a Z value of 1. The value of Z, whether greater or lesser than 1, delineates the disparity between ideal and real gas behavior. The extent of gas nonideality is shown by the disparity between one and Z. Pressure and temperature induce departures from ideal behavior in a real gas. Employing pressure and temperature to analyse the compressibility curves of the gases outlined below.
Effect of Pressure Variation on Deviations from Real Gas
The compressibility factor, Z, for H2, N2, and CO2 at constant temperature is displayed against pressure in the graph below.
At very low pressure, Z is nearly equal to one for all of these gases. At low pressures (up to 10 atm), real gases behave nearly flawlessly. H2 shows a consistent increase in Z (from Z=1) as the pressure increases. Consequently, at all pressures, the H2 curve is higher than the ideal gas curve. Z initially drops (Z1) for N2 and CO2, reaches a minimum, and then rises with increased pressure (Z>1). CO2 has the biggest dip in the curve since it is the gas that liquefies the easiest.
Effect of Temperature on Deviations from Real Gas
Plots of Z or PV/RT vs P for N2 at different temperatures are displayed in the graph below.
The graphs’ form indicates that the departures from ideal gas behaviour decrease with increasing temperature. The curve has a negative slope and a large dip at lower temperatures. Z<1 in this case. The curve’s dip gets less as the temperature rises. The minimum of the curve disappears at a specific temperature and stays horizontal across a broad pressure range. Boyle’s law is met at this temperature because PV/RT is almost equal. Boyle’s temperature so refers to the gas’s temperature. Every gas has a unique Boyle temperature; for example, N2 has a Boyle temperature of 332K.
Note:
The deviation from optimal behaviour increases as the gas gets closer to the liquefaction point.
At low pressures and relatively high temperatures, real gases behave almost perfectly, and the ideal-gas equation is followed.
At sufficiently high pressures and low temperatures, a real gas deviates significantly from ideality, rendering the ideal-gas equation invalid.
Watch Complete Lecture in Urdu/Hindi for comprehensive understanding
Download Complete Notes Below
Proudly Powered By




Leave a Comment