Arrhenius Equation
The Arrhenius equation explains how the rate of a chemical reaction depends on temperature and activation energy. It is written as:
k = A e^(-Ea/RT)
Temperature dependency of reaction rate
Activation energy is relevant. According to collision theory, a reaction proceeds when reactant molecules collide with one another. The threshold energy is the minimum energy required for colliding molecules to achieve an effective collision.
The activation energy is the minimum extra energy required for reactant molecules to reach the threshold energy level.
Threshold energy = Activation energy + Energy possessed.
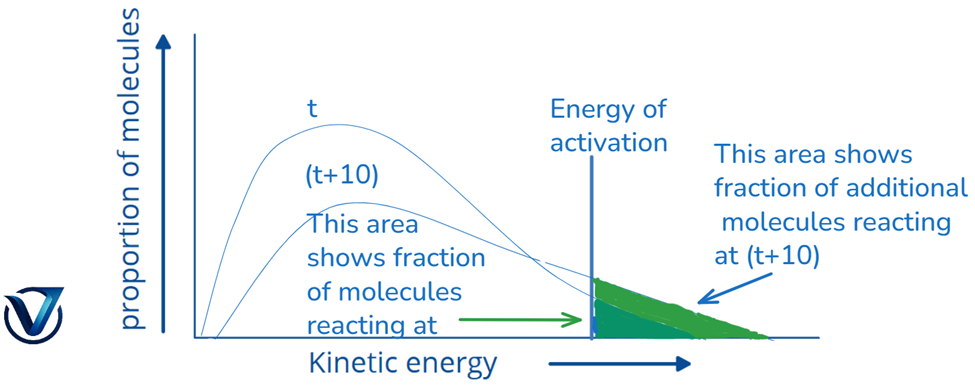
The rate constant roughly doubles for a chemical reaction when the temperature increases by 10 degrees Celsius.
Temperature coefficient =
Rate constant at T + 10° / Rate constant at T
Explanation:
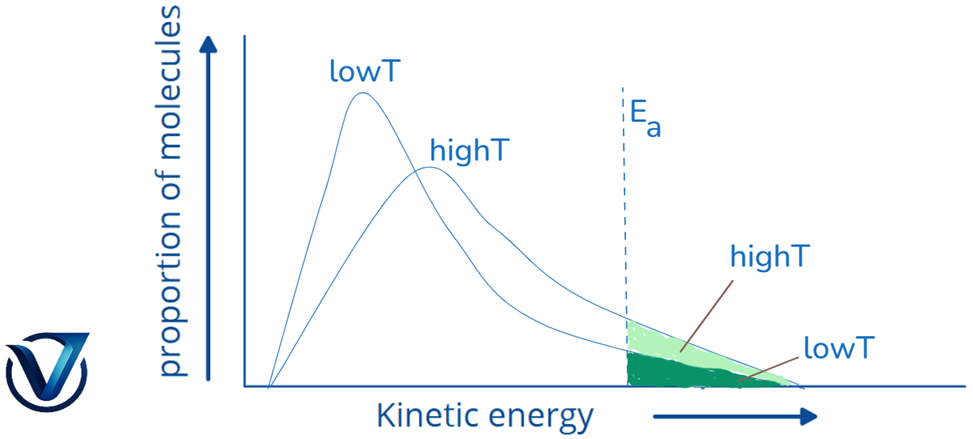
A graph such as the one illustrated is produced when the fractions of molecules are plotted against their associated kinetic energies at a designated temperature. The apex of the curve signifies the most probable kinetic energy, reflecting the kinetic energy held by the majority of molecules.
As the temperature rises
- The curve’s maximum moves to a higher energy value, signifying a rise in the most probable kinetic energy.
- The curve changes to the right, suggesting that there are more molecules with very high energies.
Given that the overall probability must equal one, the area beneath the curve stays invariant. At (t + 10), the area representing the percentage of molecules possessing energy equal to or above the activation energy doubles, hence causing the reaction rate to double.
Arrhenius equation
The Arrhenius equation quantitatively elucidates the temperature dependence of the rate of a chemical reaction.
k = Ae(-Ea/RT)
where:
- K = rate constant
- A = frequency factor
- Ea = activation energy
- R= gas constant
- T= absolute temperature
The Arrhenius factor, referred to as the frequency factor or pre-exponential factor, is denoted by A. Ea represents the activation energy in joules per mole, while R signifies the gas constant.
The proportion of molecules with kinetic energy exceeding Ea is denoted by the factor e^(-Ea/RT).
The Arrhenius equation indicates that increasing the temperature or reducing the activation energy enhances the reaction rate and exponentially increases the rate constant.
Considering both sides of the equation’s natural logarithm: ln k = -(Ea/RT) + ln AA linear graph with a slope of -Ea/R and an intercept of ln A is obtained when plotting ln k against 1/T.
At temperature T1, the equation is
lnk1 = Ea/RT1 + ln A.
At temperature T2, the equation is
lnk2 = Ea/RT2 + ln A.
In a specific response, A remains constant.
The rate constants for temperatures T1 and T2 are denoted as k1 and k2, respectively.
From the equation,
ln k₂ – ln k₁ = (Eₐ/RT₁) – (Eₐ/RT₂) ln (k₂/k₁) = (Eₐ/R) ((1/T₁) – (1/T₂))
log k₂/k₁ = (Eₐ/2.303R) × ((1/T₁) – (1/T₂))
log(k₂/k₁) = (Eₐ / 2.303R) × ((T₂ – T₁) / (T₁T₂))
Graphical representation of the impact of temperature on Arrhenius Equation
It has been established that the average kinetic energy of molecules is related to temperature. A bimolecular reaction, as per collision theory, happens solely when the reacting molecules collide with sufficient kinetic energy and appropriate orientation.
The proportion of molecules with kinetic energy equal to or above Ea at a specific temperature may result in the formation of the product. As the temperature escalates, the fraction of molecules possessing energies equal to or exceeding (>= Ea) increases. Consequently, the response rate would escalate. Graphing a subset of molecules with specific kinetic energy against kinetic energy for two different temperatures, T1 and T2, demonstrates this concept.
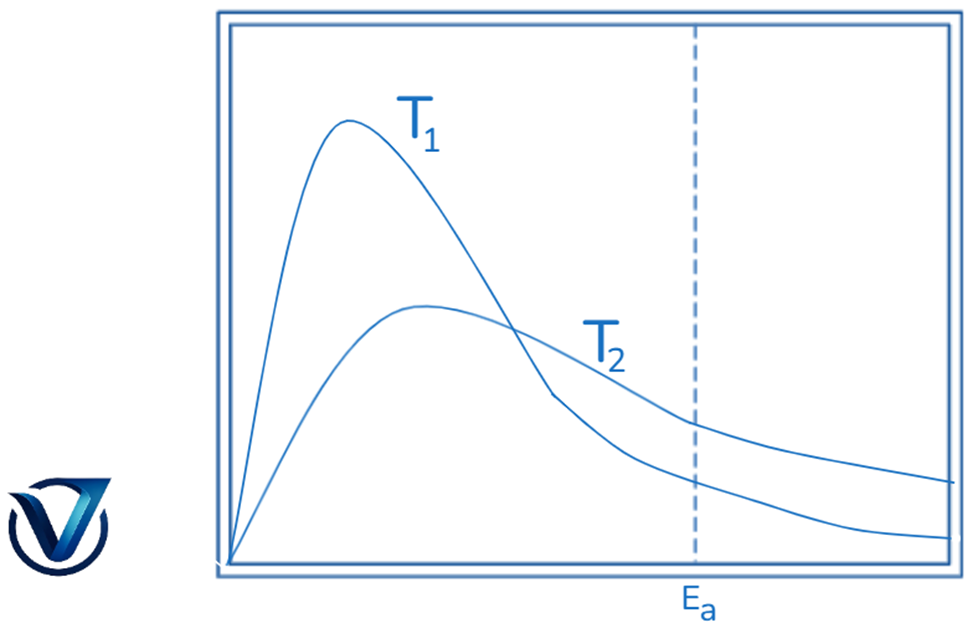
The quantity of molecules possessing certain amounts of kinetic energy is proportional to the area beneath the curve. At T1 and T2, the entire region remains identical. The proportion of molecules with kinetic energy exceeding Ea at temperatures T1 and T2 is depicted by the areas (a) and (b). This indicates that when the temperature increases, the proportion of molecules with energies above Ea also increases. Consequently, the response rate accelerates.
Use of Catalyst
A catalyst is a substance that accelerates the rate of a process without experiencing any permanent chemical alterations. A catalyst forms temporary bonds with reactants to generate an intermediate complex, as per this idea. This compound undergoes decomposition over time, yielding products and the catalyst.
A catalyst reduces the potential energy barrier by diminishing the activation energy. Consequently, the catalyst provides an alternate reaction route. Catalyse a high number of reactants using a low amount of catalyst. The Gibbs energy remains unaffected by a catalyst. A catalyst can just facilitate spontaneous reactions, excluding non-spontaneous reactions. Moreover, a catalyst does not modify the equilibrium constant; instead, it facilitates a more rapid achievement of equilibrium.
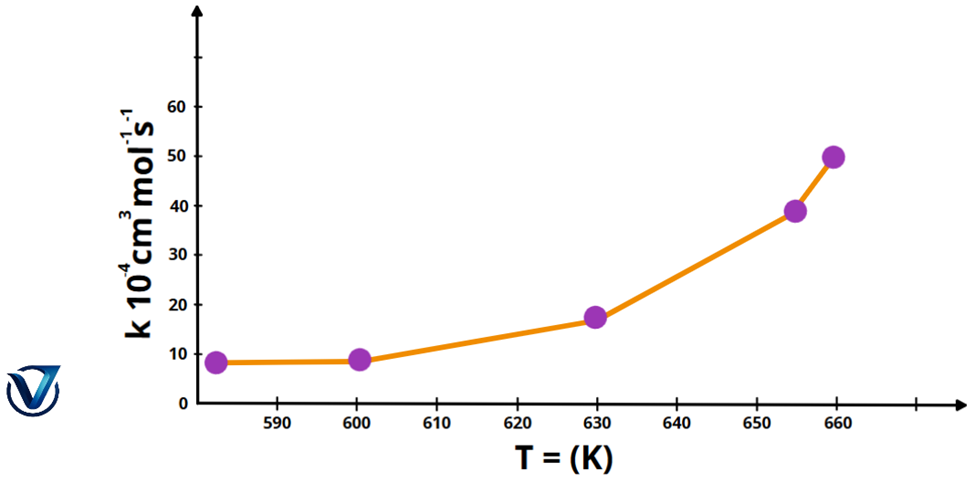
Graph of the Arrhenius Equation
We will graph the Arrhenius equation using the example of the decomposition process of nitrogen dioxide. The X-axis will denote the absolute temperature T in Kelvin, while the Y-axis will indicate the rate constant k. First, we must articulate the decomposition reaction for nitrogen dioxide.
2NO₂ → 2NO + O₂
The graphic below illustrates the breakdown of the mentioned reaction.
Watch Complete Lecture in Urdu/Hindi for comprehensive understanding
Download Complete Notes Below
Proudly Powered By




Leave a Comment