Hittorf’s rule relates to the movement of ions in an electrolyte during electrolysis. It states that the change in concentration of an ion near an electrode is proportional to the amount of electric charge passed.
The transport number (or transference number) is the fraction of total current carried by a particular ion in an electrolyte. It helps quantify ion contributions to conductivity and is essential in electrochemistry for understanding ion transport and cell efficiency.
Ion mobility (Hittorf’s Rule)
- The electric current causes ions to flow to the electrodes that are oppositely charged. However, the speeds aren’t always the same.
- The decrease in the concentration of these ions at the anode will be directly correlated with the speed at which a cation moves away from the anode. Likewise, an anion’s speed.
- After conducting an experimental study of these changes, Hittorf developed a general guideline known as Hittorf’s Rule. It says that:
The rate at which an ion moves away from an electrode determines how much concentration is lost around it.
The following scheme serves as an example of Hittorf’s Rule.
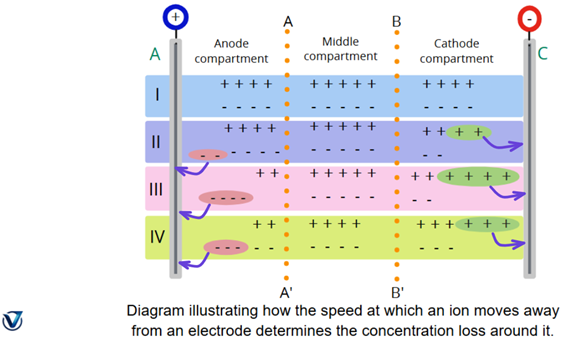
Anode A and cathode C are shown in Fig. The cell is divided into three compartments: the anode compartment, the middle compartment, and the cathode compartment by two hypothetical planes, AA’ and BB’. An anion is represented by the sign (–), whereas a cation is represented by the sign (+).
Watch Full Lecture in Urdu/Hindi
Watch full Lecture in English
The loss of concentration around any electrode is proportional to the speed of the ion moving away from it.
Hittorf’s Rule may be illustrated by the following scheme.
In the figure, A represents the anode and C the cathode. The imaginary planes AA′ and BB′ divide the cell into three compartments: anode, middle, and cathode. The (+) sign indicates cations, while the (–) sign denotes anions.
Before electrolysis begins, the cell contains 13 ion-pairs: 4 in each of the outer compartments and 5 in the middle compartment (Position I). Now, let us consider the following cases:
(i) Let the anions alone be capable of movement
If, for example, two anions move toward the anode, the arrangement changes as shown in Position II. Although the cations remain stationary, two anions and two cations are discharged, maintaining balance. The concentration in the anode compartment remains unchanged, but in the cathode compartment, it decreases by two ion-pairs.
(ii) Let the anions and cations move at the same rate
When two ions of each type move toward their respective electrodes, the arrangement becomes as shown in Position III. Four ions (two cations and two anions) are discharged, keeping the number equal. The concentrations in both the anode and cathode compartments decrease equally—by two ion-pairs each.
(iii) Let the cations move at twice the speed of the anions
In this case, as cations move toward the cathode, one anion enters the anode compartment, as shown in Position IV. A total of three ions (anions and cations) are discharged equally. While the cathode compartment loses one ion-pair, the anode compartment loses two. These observations show that ions are always discharged in equivalent amounts at both electrodes. However, differences in ion concentration arise due to the varying speeds of anions and cations. Thus, the decrease in concentration around an electrode is directly proportional to the speed of the ion moving away from it. We can write the expression:

where the speeds of cations and anions are denoted by v+ and v–, respectively.
An important assumption in the above derivation is that discharged ions do not react with the electrode material. However, in many cases, they do combine with the electrodes rather than simply depositing. This can lead to an increase in ion concentration around the electrode instead of a decrease.
Download Complete Notes Below
Proudly Powered By




Leave a Comment