An equation of state is a mathematical formula used in thermodynamics and physics that explains how a physical system behaves in terms of its thermodynamic characteristics, such as temperature, pressure, and volume. In a number of disciplines, including chemistry, materials science, aerospace engineering, and energy production, this mathematical model enables engineers and scientists to forecast and analyze a system’s behavior.
The behavior of a system according to various circumstances is described by equations of state. They can be used, for instance, to forecast how a gas will behave at various pressures and temperatures or to ascertain the characteristics of a liquid when it is heated or cooled.
Types of equations of state
Ideal gas law
The ideal gas law is a fundamental equation of state that characterizes the behavior of gases under low pressure and elevated temperature conditions. The ideal gas law correlates the pressure, volume, and temperature of a gas sample.
The ideal gas law can be employed to calculate the volume of a gas given temperature and pressure.
PV=nRT
Here:
- P= Pressure in atm
- V= Volume in liters or dm3
- T= Temperature in Kalvin
- N = Number of moles
- R= Ideal gas constant
Van der Waals equation
The Van der Waals equation is an adaptation of the ideal gas law that incorporates the intermolecular attraction and repulsive forces among gas molecules. This equation is frequently employed to characterize the behavior of gases under moderate pressures and temperatures, offering a more precise representation than the ideal gas law in numerous situations.
The Van der Waals equation considers the volume of gas molecules and the intermolecular attractive forces. This renders it more precise than the ideal gas law in scenarios when gas molecules are in close proximity and interact with one another.
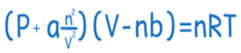
Here, ‘a’ and ‘b’ are Van der Waals constants and contain positive values. The constants are characteristic of the individual gas. When gas is ideal, or it behaves ideally, then both the constants will be zero. Generally, a constant helps in the correction of the intermolecular forces, while the b constant helps in making adjustments for the volume occupied by the gas particles.
Virial equation of states
The behavior of both liquids and gases over a larger range of pressures and temperatures can be described by the more complicated Virial equation of state. The Virial equation provides a more realistic representation of complex systems because it takes into consideration the interactions of three or more molecules in addition to the pairwise interaction of gas molecules.
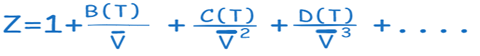
The virial equation of states are these and related equations, and the coefficients a(T), b(T), c(T), and so forth that are only dependent on temperature are referred to as Virial coefficients. Statistical mechanics can be used to theoretically or experimentally determine these coefficients.
In the study of statistical mechanics and thermodynamics, the Virial equation is frequently employed. It is especially helpful in comprehending how gases and liquids behave at low temperatures and high pressures.
Redlich-Kwong equation
A popular equation of states for explaining the behaviors of non-ideal gases and liquids is the Redlich-Kwong equation. This formula takes into consideration the behaviour of polar molecules, intermolecular forces among molecules, and other complex interactions.
Chemical engineers frequently employ the Redlich-Kwong equation, especially when designing and optimizing industrial operations involving gases and liquids.
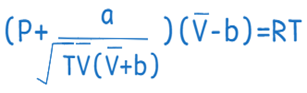
Peng-Robinson equation
Another well-known equation of states that is frequently utilized in the petroleum sector for simulating and modelling different hydrocarbon mixes is the Peng-Robinson equation. When estimating the phase behavior of complicated mixtures at high temperatures and pressures, this equation is especially helpful.
The size, shape, and intermolecular forces of the molecules in the mixture are all taken into consideration by the Peng-Robinson equation. It is frequently employed in the planning and improvement of procedures for the extraction and transportation of gas and oil.
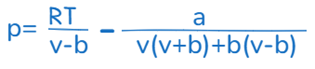
Applications of equations of state
- Phase behavior prediction: Researchers can forecast a substance’s behavior under a variety of variables, including temperature, pressure, and composition, using equations of state and other thermodynamic models.
- Calculating thermodynamic properties: Entropy, enthalpy, fugacity, and many other thermodynamic properties can be computed using equations of state, help in the development and optimization of numerous processes.
- Chemical reaction modeling: By coupling EOS with reaction kinetics, researchers can simulate how pressure and temperature shifts affect equilibrium positions.
- This is especially important in ammonia synthesis (Haber process) or methanol production, where reaction yields depend heavily on thermodynamic conditions.
- Applications in engineering and industry: from creating novel materials for energy storage to creating chemical reactors. Equations of state enable engineers to create and optimize procedures while reducing expenses and increasing efficiency by giving a mathematical representation of a system’s behavior.
Equation of States Lecture in Urdu/Hindi
Download Complete Notes Below
Proudly Powered By




Leave a Comment