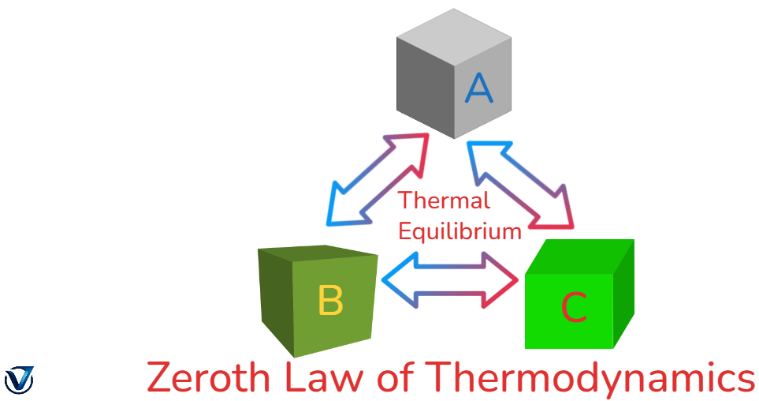
The Zeroth Law of Thermodynamics states that if two bodies are in thermal equilibrium with a third body, then those two bodies are also in thermal equilibrium with one another. This signifies that if system A is in thermal equilibrium with system B, and system C is also in thermal equilibrium with system B, then systems A and C are in thermal equilibrium with each other.
This law got name because all preceding laws were formulated and named prior to it. Subsequent to the formulation of this rule, it became evident that it had greater utility than its predecessors, thereby earning the designation of the zeroth law of thermodynamics.
Applications of Zeroth Law of Thermodynamics
The thermometer exemplifies the most prevalent use of the Zeroth law of thermodynamics. The zeroth law may be readily proven with a mercury thermometer, as the tube’s cross-sectional area remains constant while the mercury level fluctuates with temperature variations. The fluctuation in mercury height signifies a change in temperature. Various types of thermometers are employed to measure distinct amounts. A constant gas volume thermometer measures pressure; a constant pressure gas thermometer measures volume; an electrical resistance thermometer measures resistance; and a mercury-in-glass thermometer measures length.
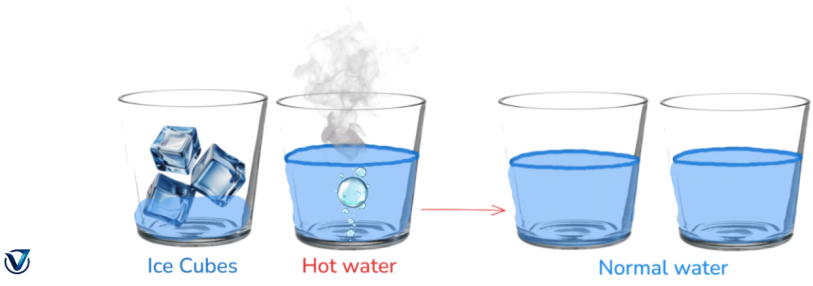
· Glasses containing water at varying temperatures: An additional example may be effectively illustrated by presenting two glasses of water, one filled with cold water and the other with warm water. Both glasses will reach room temperature to achieve thermal equilibrium.