Van der Waals Equation
The Van der Waals equation is a state equation that characterises the behaviour of actual gases, including the volume of gas molecules and the intermolecular attraction and repulsive interactions. The equation is named for Dutch scientist Johannes Diderik van der Waals, who introduced it in 1873.
Variable Definitions
Core Assumptions of Real Gases
The Van der Waals equation is predicated on two assumptions regarding real gases:
1. Significant Molecular Volume: The volume of gas molecules in actual gases is significant relative to the volume of the container. This indicates that the true volume filled by the gas is marginally smaller than the volume of the container.
2. Intermolecular Forces: In actual gases, attractive and repulsive interactions exist between gas molecules. Attractive interactions diminish the gas pressure, while repulsive interactions elevate it.
The Van der Waals equation considers these aspects and offers a more precise depiction of real gas behaviour, especially under high pressures and low temperatures, where the ideal gas law fails.
Volume Correction
The volume of a gas refers to the quantity of unoccupied space within a container that allows molecular movement. While the volume V of an ideal gas corresponds exactly to the volume of its container, Van der Waals conceptualised molecules in a true gas as hard spherical entities with a definitive volume.
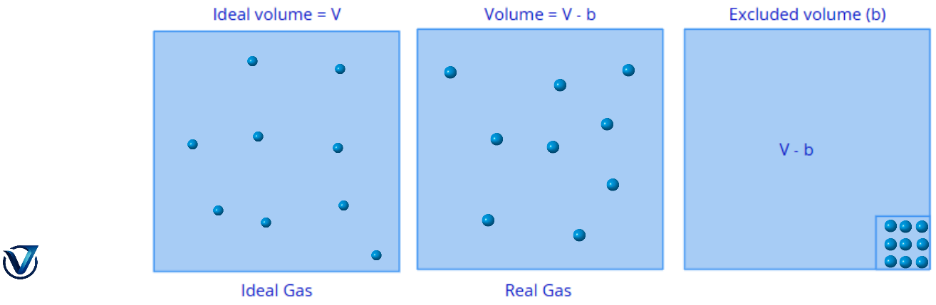
The volume of a real gas is considered the ideal volume minus the amount occupied by the gas molecules themselves. Unlike ideal gases, real gas molecules possess a definitive volume that restricts the space available for free movement.
The excluded volume, denoted as b, is a constant and distinct value for each specific gas.
Pressure Correction and Intermolecular Forces
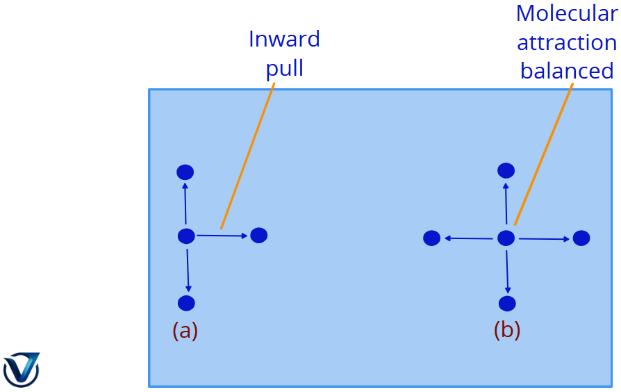
In a real gas, the pressure exerted is influenced by the attractive forces between molecules. This differs significantly from the ideal model where these forces are neglected.
- A molecule deep within a gas is subjected to attractive forces from surrounding molecules on all sides. These forces negate one another, resulting in no net pull.
- A molecule about to strike the vessel’s wall is attracted solely by molecules on one side (the interior).
As a result, the molecule impacts the wall with diminished force. This leads to the actual gas pressure, P, being inferior to the ideal pressure, Pideal.
Mathematical Derivation of Ideal Pressure
If the actual pressure P is less than the ideal pressure Pideal by a factor of p, we use the following relationship:
Pideal = P + p
This correction accounts for the internal “contrived” entry of molecules as they approach the boundaries, providing a precise depiction of real gas behavior under various physical conditions.
Derivation of the Pressure Correction Factor (p)
Van der Waals derived the value of the pressure correction quantity p (often denoted as P’) by considering the density of the molecules at the walls and within the bulk of the gas.
P’ ∝ (n/V) • (n/V)
P’ = a(n/V)2
Where n represents the total number of gas molecules in volume V, and a signifies the proportionality constant specific to the gas.
The Van der Waals Equation for n Moles
Substituting the altered pressure and volume values into the ideal gas equation (PV = nRT) yields the complete real gas equation:
The Equation for 1 Mole of Gas
For specialized calculations where n = 1, the equation simplifies to:
Units of Van der Waals Constants
Mastering the units is essential for dimensional analysis in competitive chemistry exams:
atm • lit2 • mol-2
litre • mol-1


