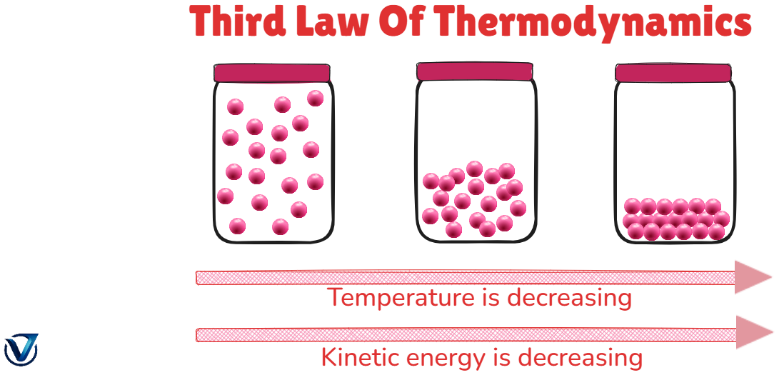
The third law of thermodynamics asserts that when temperature nears absolute zero, the entropy of a system converges to a constant value. At absolute zero, the entropy of a pure crystalline material is zero. If the ideal crystal possesses a singular state of little energy, this statement is valid.
Implementation of the Third Law of Thermodynamics
The use of the third law of thermodynamics facilitates the computation of absolute entropy at any temperature ‘T,’ relying on the heat capacity measurements of the substance.
The third law of thermodynamics has two principal applications, outlined below.
- The third law of thermodynamics is applied. It assists in determining if compounds are pure crystalline or not.
- It assists in determining the absolute entropy of substances at a certain temperature.
The third law of thermodynamics pertains to fully crystalline substances to determine their purity as crystalline materials. It delineates the entropy of a completely crystalline material, which may be zero at absolute zero temperature (0 Kelvin). If the substance lacks complete crystallinity, its entropy will not equal zero at 0 Kelvin.
Inconsistency with the other principles of thermodynamics
The third law of thermodynamics identifies absolute zero as a state, while the second rule of thermodynamics asserts that temperature can never reach zero. According to the second law of thermodynamics, heat cannot spontaneously transfer from a colder body to a hotter body. If a system attempts to attain absolute zero, it inherently tends to extract heat from its external environment, should this occur. Consequently, it will never attain absolute zero.
The first law is that energy cannot be generated or destroyed. In this scenario, thermal energy must be extracted from the external environment, so precluding the possibility of the system attaining absolute zero.
The significance of the third law of thermodynamics
1. It assists in the computation of thermodynamic characteristics.
2. It aids in quantifying chemical affinity. This is referred to as the Nernst theorem.
3. It elucidates the behavior of solids at extremely low temperatures.
4. It assists in the analysis of chemical and phase equilibrium.


