Measurement of Electrolytic Conductance
Conductance tells us how easily electricity passes through a solution. It’s the reciprocal of resistance, meaning if resistance is high, conductance is low, and vice versa.
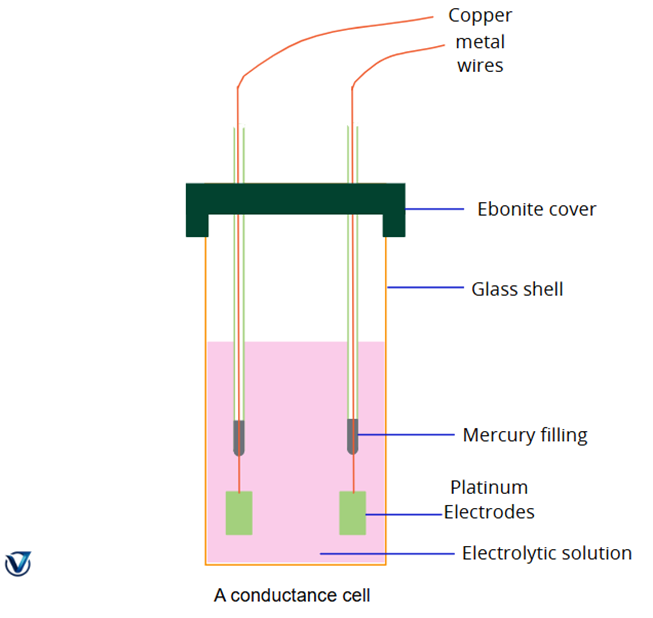
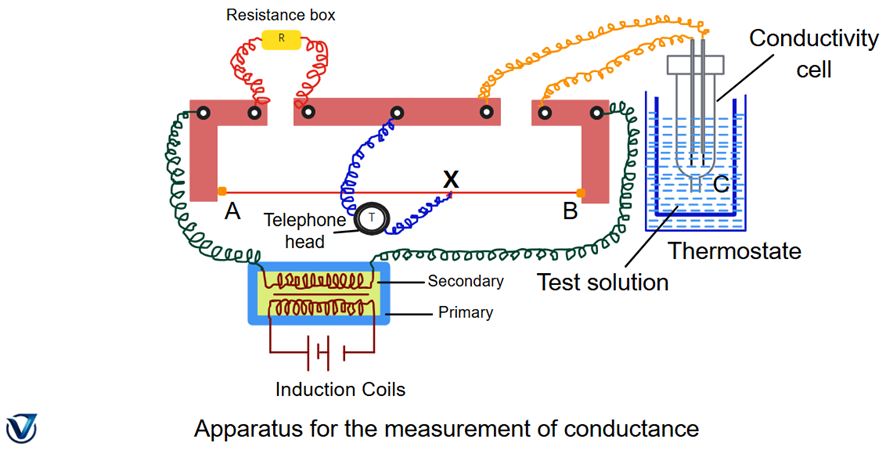
To measure conductance, a Wheatstone bridge setup is used. The solution is placed in a conductance cell fitted with platinum electrodes coated with platinum black (to prevent polarization and ensure accurate readings). The electrodes are connected to the circuit through mercury contacts and copper wires. A headphone replaces the galvanometer to detect the balance point (minimum sound indicates balance). The resistance box (R), sliding contact (H), manganin wire (AB), and induction coil (I) complete the circuit. Once resistance is measured, conductance (G) is calculated as:

From this, we can find:
- Specific conductance (k) — conductance of 1 cm³ of solution.
- Equivalent conductance (Λ) — conductance per gram-equivalent of solute.
Here, N is the gram-equivalent.
- Molar conductance (Λₘ) — conductance per mole of solute.
Here, M is the gram-mole of the electrolyte.
The cell constant (x =1/A) depends on electrode spacing and area. It’s usually determined indirectly using a standard KCl solution whose conductance is known.
Think of conductance as how easily electricity “flows” through a liquid.
• If ions move freely, conductance is high.
• If movement is restricted, conductance is low.
In the lab, the Wheatstone bridge acts like a balance scale for electricity. When the headphone sound fades, it means the bridge is balanced — that’s the point where resistance can be measured accurately.
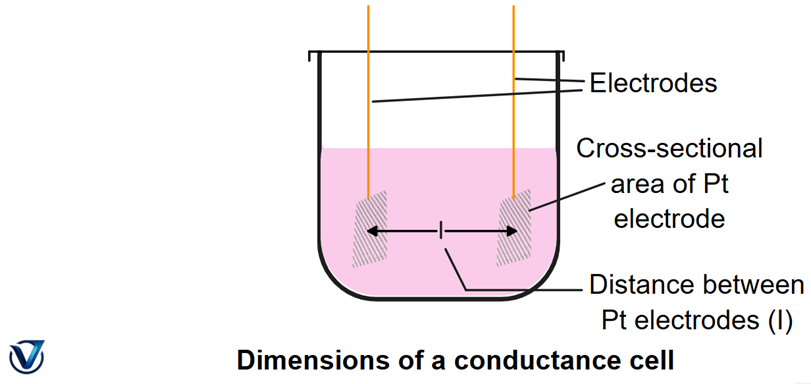
The cell constant is like a fingerprint of the conductance cell — it depends on how far apart the electrodes are and how big they are. Because measuring these dimensions directly is tricky, that’s why scientists use a known KCl solution to calibrate the cell.
Once the cell constant is known, we can easily find the conductance of any unknown solution by comparing it to the standard.


