Fugacity and activity explained with clear concepts, definitions, and significance in thermodynamics and chemical engineering. This guide covers their roles in real gas behavior, non-ideal solutions, equilibrium calculations, and phase transitions, helping students understand how these parameters correct ideal models for accurate predictions in practical chemical and physical systems.
Activity
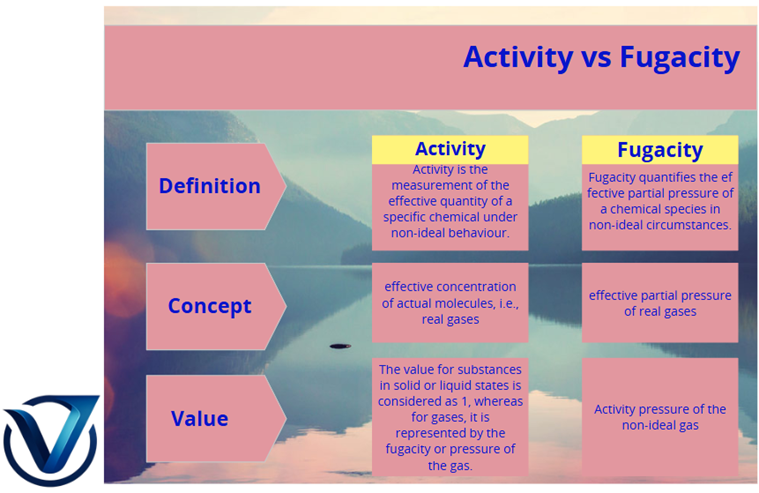
Activity quantifies the effective concentration of a chemical species in non-ideal conditions. The notion of activity was formulated by the American chemist Gilbert N. Lewis. Activity is a dimensionless parameter. The activity of a certain chemical is contingent upon its standard state. The value assigned to substances in solid or liquid states is considered to be 1. In the context of gases, activity pertains to the effective partial pressure, defined as the fugacity or pressure of the gas under consideration. Moreover, the activity is contingent upon the following factors:
- Temperature
- pressure
- Constitution of the amalgamation, etc.
The environment influences the behavior of a chemical species. The gas molecules in non-ideal circumstances tend to interact, either attracting or repelling one another. Consequently, the activity of a molecule or ion is influenced by the chemical species in its vicinity.
Fugacity
Fugacity quantifies the effective partial pressure of a chemical species in non-ideal circumstances. The fugacity of a certain chemical species, such as a real gas, is equivalent to the pressure of an ideal gas at the same temperature and molar Gibb’s energy as the real gas. Fugacity can be ascertained through experimental methods or alternative models, such as the Van der Waals gas, which more accurately represents real gases compared to ideal gases.
The fugacity coefficient serves as the connection between the pressure of a real gas and its fugacity. It can be represented by the sign ϕ. The connection is expressed as ϕ = f/P.
In this context, f denotes fugacity, whereas P represents the pressure of the actual gas. For a perfect gas, pressure and fugacity are equivalent. Consequently, the fugacity coefficient for an ideal gas is unity.
Watch Complete Lecture in Urdu/Hindi for comprehensive understanding
Download Complete Notes Below
Proudly Powered By




Leave a Comment