In an isolated system, the second rule of thermodynamics states that entropy perpetually grows. Any isolated system naturally evolves towards thermal equilibrium or the condition of maximal entropy. The entropy of the cosmos is perpetually expanding and never diminishing.
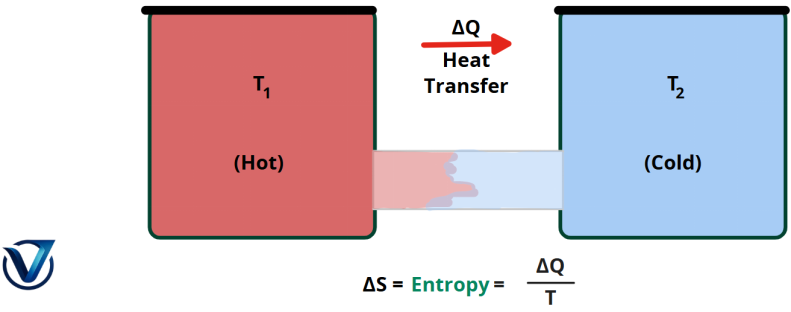
The second law of thermodynamics is theoretically represented as
ΔSuniversal > 0
Here,
ΔSuniversal > 0is a change in the universe’s entropy.
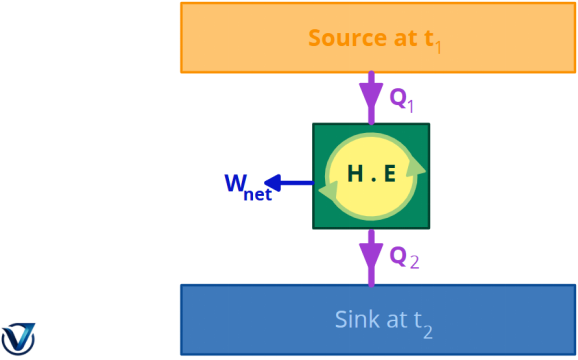
Kelvin-Planck Statement of the Second Law of Thermodynamics
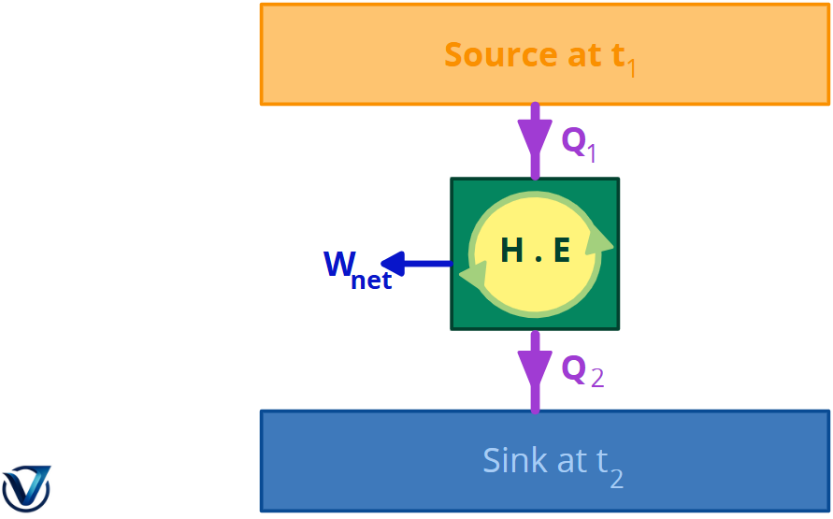
A heat engine cannot generate work in a whole cycle if it simply exchanges heat with bodies at a single, constant temperature.
No engine can transform all of the heat from the source into work without wasting any heat. This indicates that a sink is required to get continuous work.
The subsequent illustration illustrates the operation of the heat engine.
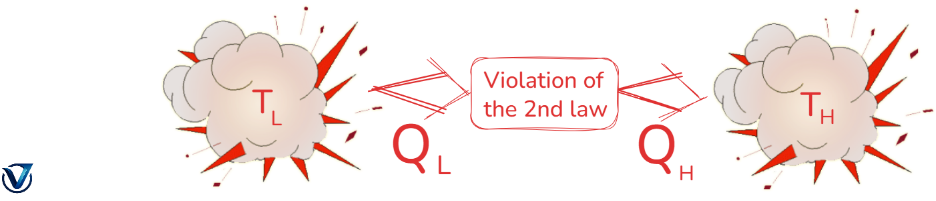
Clausius Statement of Second Law of Thermodynamics
Technology that can transfer heat from a colder body to a warmer body without wasting energy is unattainable. Put another way, unless the compressor is powered by an external source, the refrigerator will not function. Heat pumps and freezers use Clausius’s claim.
The statements made by Clausius and Kelvin are comparable, meaning that a device that violates Clausius’s statement will also violate Kelvin’s statement, and vice versa.
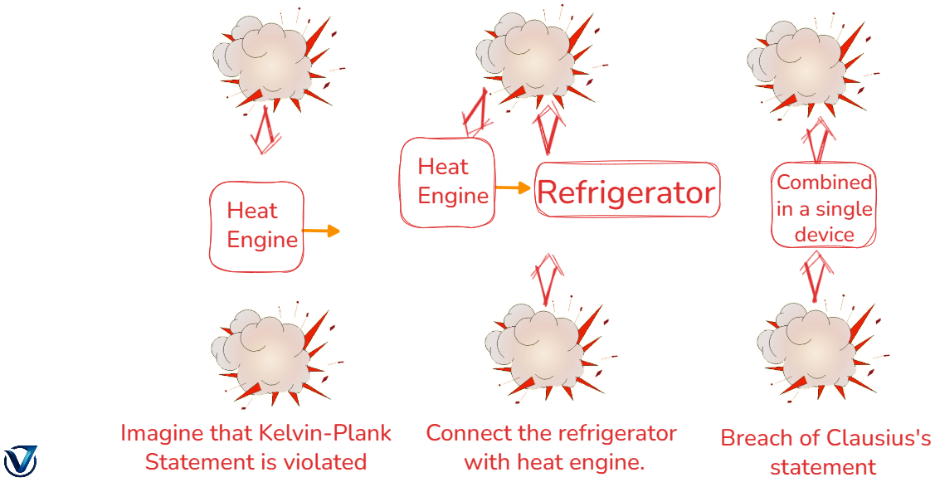
Second Kind Perpetual Motion Machine (PMM2)
A device that does work while engaging with a single heat reservoir is referred to as a perpetual motion machine of the second sort (PMM2). A device that contravenes the second rule of thermodynamics is a perpetual motion machine of the second sort.
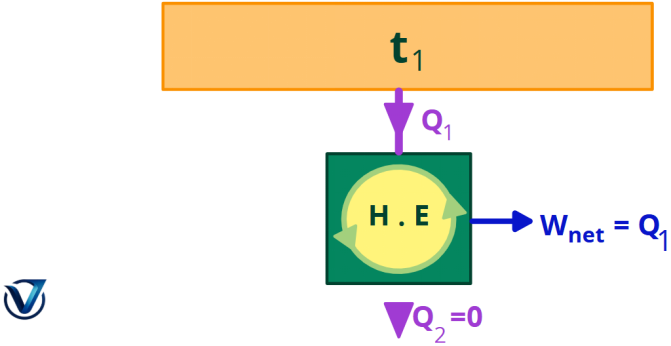
Consequently, a heat engine must engage with a minimum of two thermal reservoirs at disparate temperatures to generate work in a cycle. As long as there is a temperature differential, mechanical work may be generated. The heat engine will generate work until the temperatures of the two substances with finite heat capacity are equalized.
Applications of the Second Law of Thermodynamics
Applications and Uses of the Second Law of Thermodynamics
- The law stipulates that heat invariably transfers from a warmer body to a colder body. This regulation encompasses all heat engine cycles, including Otto and Diesel, as well as all working fluids utilized in these engines. This law has propelled advancements in modern autos.
- Refrigerators and heat pumps employing the Reversed Carnot Cycle exemplify the use of this principle. External work is required to transfer heat from a body at a lower temperature to a body at a higher temperature. In contrast to the Reversed Carnot Cycle, which employs effort to transfer heat from a lower-temperature reservoir to a higher-temperature reservoir, the original Carnot Cycle utilizes heat to generate work
.Is it feasible to preserve Qout?
In a steam power station, the condenser is the apparatus that expels substantial amounts of waste heat to rivers, lakes, or the environment. One can inquire whether it is feasible to remove the condenser from the plant to conserve all that wasted energy. The response to this inquiry is unequivocally negative, as the absence of a heat rejection process in a condenser precludes the completion of the cycle. Cyclic equipment, such as steam power plants, cannot operate continually unless the cycle is fulfilled. This is illustrated subsequently with the aid of a basic heat engine.
Examine the basic heat engine seen in following Fig. which is employed to elevate weights. The apparatus comprises a piston-cylinder mechanism including two sets of stops. The working fluid is the gas housed within the cylinder. The initial gas temperature is 30°C. The piston, burdened with weights, is positioned atop the lower stops. 100 kJ heat is delivered to the gas within the cylinder from a source at 100°C, resulting in its expansion and elevating the loaded piston until it reaches the upper stops, as seen in the figure. The load has been removed, and the gas temperature is recorded at 90°C.
The work performed on the load during this expansion phase corresponds to the augmentation of its potential energy, namely 15 kJ. Even in optimal conditions (weightless piston, absence of friction, no thermal losses, and quasi-equilibrium expansion), the heat imparted to the gas exceeds the work performed, as a portion of the heat is utilized to elevate the gas’s temperature.
We now address the inquiry: Is it feasible to transmit 120 kJ of surplus heat at 80°C back into a reservoir sustained at 110°C for reutilization? Under optimal conditions, this procedure would indicate a heat engine functioning at 100% efficiency. Nevertheless, the response is negative, as the fundamental principle states that heat inherently transfers from a hotter body to a colder one, never in the other direction. Consequently, it is impractical to cool this gas from 80°C to 25°C by dissipating energy into a reservoir at 110°C. The system must be linked to a lower-temperature reservoir, around 15°C, enabling the gas to revert to its original form by expelling 120 kJ of excess energy as heat. This discarded energy is irretrievable and is accurately categorized as waste energy.
We deduce from this discussion that every heat engine must dissipate a portion of energy by transferring it to a low-temperature reservoir to function effectively.


