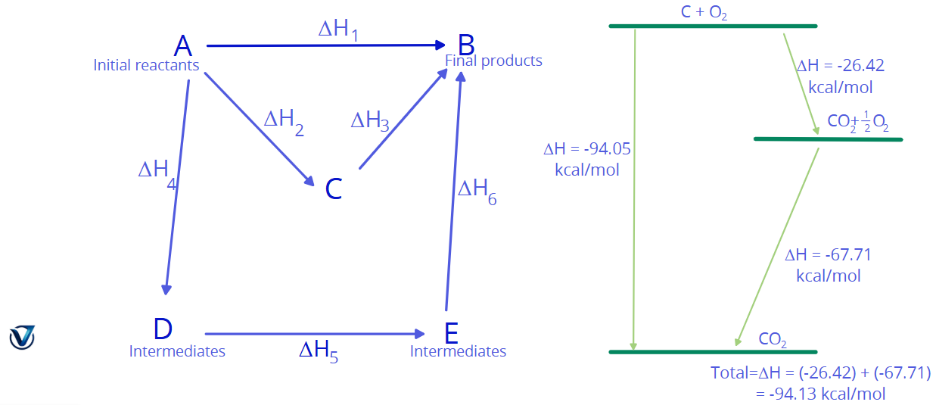
It may be stated as follows: Hess’s Law if a chemical transformation can occur through several pathways, whether in a single step or in several stages, the total heat change remains constant regardless of the technique employed to bring about the change.
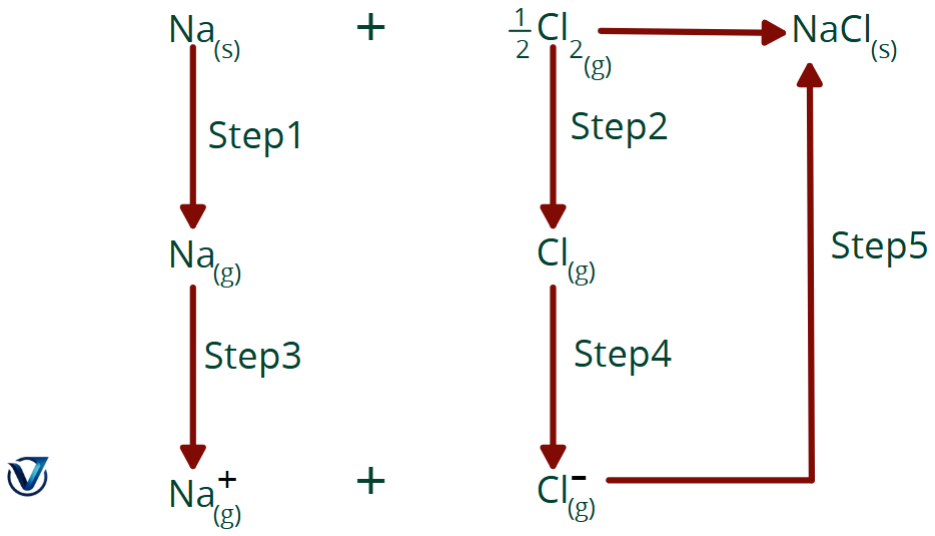
Born-Haber cycle
Enthalpy changes for direct synthesis
The enthalpy change for the direct synthesis of NaCl from sodium metal and chlorine gas is -114 kJ.
Na(s) + Cl2
NaCl(s) ΔH° = -114 kJ
Enthalpy alterations using indirect methods
The direct enthalpy change of NaCl mentioned above is equivalent to the enthalpy change of the five processes involved in the production of NaCl.
Step 1. Sublimation of sodium metal to gaseous atoms.
Na(s) Na(g) ΔH°1 = +108kJ
Step 2. Dissociation of chlorine molecules into chlorine atoms. The enthalpy of dissociation is 121 kJ/mol for chlorine.
Cl2(g)
Cl (g) ΔΗ°2 = +121 kJ
Step 3. Conversion of gaseous sodium to sodium ions by electron loss. The enthalpy of ionisation, or ionisation energy, is 495 kJ per mole of sodium atoms.
Na(g) Na+(g) + e– ΔΗ°3 = +495kJ
Step 4. Chlorine atoms get an electron to generate chloride ions. The energy released corresponds to the electron affinity of chlorine, quantified as
Cl + e– Cl–(g) ΔΗ°4 = – 348kJ
Step 5. Sodium and chloride ions combine to create a crystal lattice. The energy in this operation is the inverse of lattice energy.
Na+(g) + Cl– (g) → NaCl(s) ΔΗ°5 = – (lattice energy)
The lattice energy may be determined by equating the enthalpy of creation of NaCl, -411 kJ, to the cumulative enthalpy changes of the five phases.
ΔΗ°1 + ΔΗ°2 + ΔΗ°3 + ΔΗ°4 + ΔΗ°5 = – 411kJ
108 kJ + 121 kJ + 495 kJ – 348 kJ – (lattice energy) = -411 kJ
Solving this equation yields
Lattice energy: +787 kJ/mol
The sequence of transformations seen in the aforementioned figure is referred to as the Born-Haber Cycle, named after Max Born and Fritz Haber, who developed this technique for estimating lattice energies.


