According to the first rule of thermodynamics, energy cannot be created or destroyed, but it may be transformed from one form to another. The first law of thermodynamics incorporates heat, internal energy, and work. This law states that a portion of the heat supplied to the system is employed to change the internal energy, while the remainder is utilized to execute work.
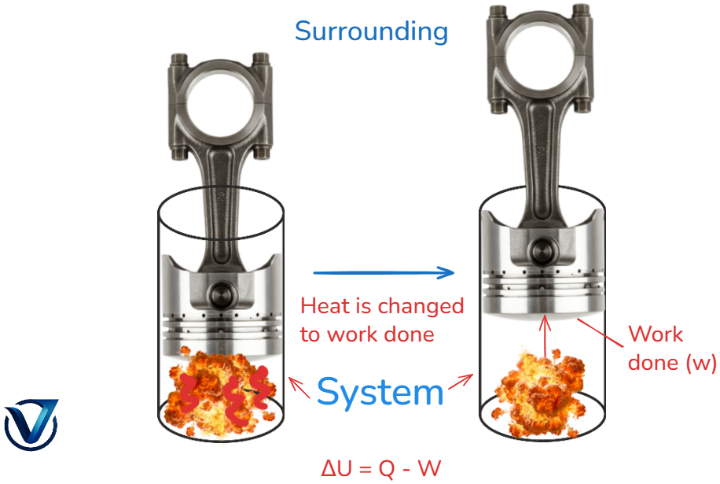
It may be mathematically written as ΔQ = ΔU + W.
Here,
• The heat exchanged is represented by ΔQ.
• The variation in internal energy is represented by ΔU.
• W denotes performed work.
The aforementioned equation may be expressed in the following manner:
ΔU = ΔQ – W
Consequently, we may infer that the quantity (ΔQ – W) is invariant regardless of the path chosen to alter the state. Moreover, the application of heat to a system generally results in an increase in internal energy, and conversely.
for an Isolated System
The work done in a closed system is the product of the applied pressure and the resultant change in volume.
W = -P ΔV
Here
• P represents the system’s constant external pressure, and
• V represents the alteration in volume.
This is termed Pressure-Volume work.
The internal energy of a system increases or decreases due to work interactions at its boundaries. When work is performed on the system, the internal energy increases; conversely, it decreases when work is performed by the system. Any thermal exchange between the system and its environment modifies the system’s internal energy.
Applications of First Law of Thermodynamics
- Energy production and utilization: The first rule of thermodynamics is employed to forecast the efficiency of energy conversion processes, such as transforming heat into mechanical work in a steam turbine or turning chemical energy into electrical energy in a battery. It is utilized to analyze engine performance and to develop more efficient and effective energy-generating technologies.
- Refrigeration and air conditioning: The first law of thermodynamics is employed to comprehend the behavior of refrigerants and to engineer refrigeration and air conditioning systems that are both efficient and effective in cooling.
- Heat transfer: The first rule of thermodynamics elucidates the dynamics of heat transfer mechanisms, including conduction, convection, and radiation. It is utilized for the design of heat exchangers and other apparatuses that facilitate the transfer of heat from one site to another.
- Chemical reaction: The first law of thermodynamics is employed to comprehend the kinetics of chemical processes and to anticipate the energy transformations that transpire throughout such reactions.
- Gaseous thermodynamics: The first law of thermodynamics elucidates the behavior of gases and forecasts the variations in temperature, pressure, and volume that transpire throughout various operations.
- Biological systems: The first rule of thermodynamics is employed to comprehend the dynamics of biological systems, including the metabolism of living beings. It is utilized to engineer gadgets and technology employed in medicine, including pacemakers and artificial organs.


