Electrolytic and Electrochemical Cells
Electrolytic and electrochemical cells are key to electrochemistry, which deals with chemical reactions involving electricity. Electrochemical cells produce electricity from spontaneous reactions, while electrolytic cells use electricity to drive non-spontaneous reactions. Understanding their difference is important in applications like batteries and electroplating.
Electrolytic cells
Electrolytic cells use an external power source to drive non-spontaneous reactions. They contain two electrodes—anode and cathode—immersed in an electrolyte. When current is applied, ions move toward the electrodes: oxidation occurs at the anode and reduction at the cathode.
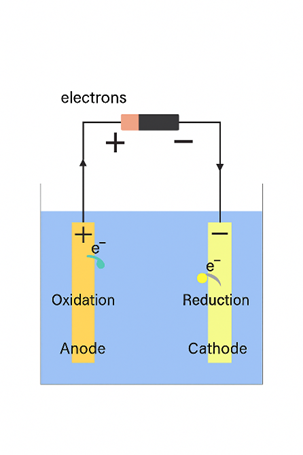
Voltaic cells generate electricity through a spontaneous chemical reaction, forming the foundation of modern batteries. However, they are not the only type of electrochemical cell. Electrolytic cells, in contrast, use electrical energy to force a non-spontaneous redox reaction to occur. This process, known as electrolysis, drives chemical changes that wouldn’t happen on their own.
The Electrolysis of Molten NaCl
The diagram below shows an idealized setup for the electrolysis of molten sodium chloride. A direct current (DC) power source is connected to two inert electrodes placed in the molten salt. In this molten state, sodium ions (Na⁺) migrate toward the negatively charged electrode (cathode), while chloride ions move toward the positively charged electrode (anode).
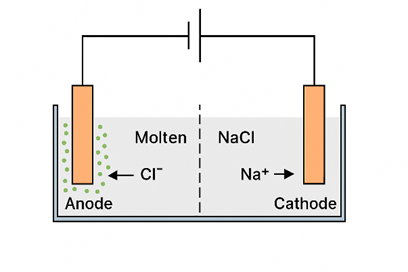
When Na⁺ ions reach the negative electrode (cathode), the applied voltage from the battery is sufficient to force them to gain electrons, resulting in the formation of sodium metal.
| Negative electrode (cathode): | Na+ + e– → Na |
Cl⁻ ions that reach the positive electrode (anode) lose electrons and are oxidized to form chlorine gas (Cl₂), which bubbles off at the electrode.
| Positive electrode (anode): | 2 Cl– → Cl2 + 2 e– |
The overall result of passing an electric current through molten sodium chloride is its decomposition into elemental sodium metal and chlorine gas.
| Electrolysis of NaCl Complete Summary: | |
| Cathode (-): | Na+ + e– → Na |
| Anode (+): | 2 Cl– → Cl2 + 2 e– |
The oxidation of Cl⁻ ions to Cl₂ requires a potential of –1.36 volts, while the reduction of Na⁺ ions to sodium metal requires –2.71 volts. Therefore, the battery must supply at least 4.07 volts to drive the overall reaction. This illustrates why the process is called electrolysis—derived from the Greek word lysis, meaning “to loosen” or “split.” Electrolysis uses electrical energy to break down a compound into its elemental components.
| 2 NaCl(l) → 2 Na(l) + Cl2(g) |
This example also highlights the key difference between voltaic and electrolytic cells. Voltaic cells harness the energy released by a spontaneous chemical reaction to perform electrical work, whereas electrolytic cells consume electrical energy to drive a non-spontaneous reaction in the reverse direction.
The dotted vertical line in the figure represents a diaphragm, which serves to prevent the chlorine gas formed at the anode from mixing with the sodium metal produced at the cathode. This separation is essential for safety and efficiency. The purpose and function of this diaphragm become clearer when examining a more detailed diagram of the commercial Downs cell, commonly used for the electrolysis of sodium chloride.
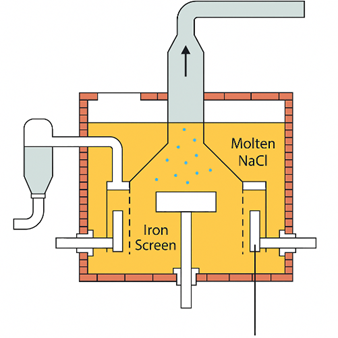
In the Downs cell, chlorine gas produced at the graphite anode rises through the molten sodium chloride and collects in a funnel at the top of the cell. Meanwhile, the sodium metal formed at the cathode, being less dense, floats upward through the molten salt and is gathered in a sodium-collecting ring, where it is periodically removed. An iron gauze screen acts as the diaphragm between the electrodes, preventing the highly reactive chlorine gas and sodium metal from coming into contact and causing a potentially explosive reaction.
The feedstock for the Downs cell is a mixture of calcium chloride (CaCl₂) and sodium chloride (NaCl) in a 3:2 mass ratio. This mixture is preferred because it melts at 580°C, significantly lower than the melting point of pure sodium chloride, which exceeds 800°C. Lowering the melting point reduces energy consumption and makes the process more efficient.
Predicting Electrolysis Reaction in Electrolytic Cells
Even if the applied voltage exceeds the theoretical requirement, four key factors determine whether electrolysis will actually proceed:
1. Overpotential: Additional voltage may be needed to overcome surface interactions at the electrodes, especially in gas-evolving reactions. For instance, hydrogen gas (H₂) often requires a 1.5 V overpotential, while platinum (Pt) may require none.
2. Multiple Possible Reactions: More than one half-reaction may occur at an electrode, leading to several possible overall cell reactions depending on which pathway is favored.
3. Nonstandard Conditions: Deviations from standard conditions—such as ion concentration or pH—can alter the required electrode potentials. For example, at 5.5 M chloride concentration, the oxidation potential changes from 1.36 V to 1.31 V. Similarly, changes in pH from the standard value affect electrode voltages.
4. Electrode Type: Inert electrodes (e.g., platinum or graphite) rely solely on species in the electrolyte to carry out reactions, while active electrodes can participate directly in oxidation or reduction.
By considering these four factors, we can more accurately predict the actual electrode and overall reactions occurring during electrolysis.
Campontents of Electrochemical Cells
On the other hand, electrochemical cells—such as galvanic or voltaic cells—produce electrical energy from spontaneous redox reactions. These cells convert chemical energy into electricity by generating a flow of electrons from the anode (where oxidation occurs) to the cathode (where reduction takes place).

Components of Electrochemical Cells
An electrochemical cell is structured to keep the oxidizing and reducing agents physically separated, while still allowing electrons to transfer between them via an external circuit. This controlled separation is essential for harnessing or supplying electrical energy through chemical reactions.
1. Anode (Where Oxidation Happens)
- The anode is the electrode where oxidation (loss of electrons) takes place.
- In a Galvanic Cell: The anode is negative because the substance at this electrode loses electrons, which then flow into the external circuit (e.g., Zn → Zn²• + 2e»).
- In an Electrolytic Cell: The anode is positive, as an external power source pulls electrons away from the electrode to drive a non-spontaneous reaction.
- Key Point: Regardless of the cell type, oxidation always occurs at the anode.
2. Cathode (Where Reduction Happens)
- The cathode is the electrode where reduction (gain of electrons) occurs.
- In a Galvanic Cell: The cathode is positive because it attracts and receives electrons from the external circuit.
- In an Electrolytic Cell: The cathode is negative, as electrons are supplied by an external power source to drive a non-spontaneous reduction.
- Example: Cu²• + 2e» → Cu(s) — reduction of copper ions to solid copper at the cathode.
- Key Point: Reduction always occurs at the cathode, regardless of the cell type.
3. Electrodes (Active vs. Inert)
- Active Electrodes: These take part directly in redox reactions and must be solid and conductive (e.g., Zn, Cu). In a Daniell cell, for example, the zinc electrode loses mass as it oxidizes, while the copper electrode gains mass as it is reduced.
- Inert Electrodes: These do not participate in the chemical reaction but serve as a surface for electron transfer. They are used when the redox species are in solution or gaseous form, such as platinum or graphite in systems involving H² gas or Fe³•/Fe²• ions.
4. Electrolyte
- A conductive ionic solution that enables the flow of charge within the electrochemical cell.
- It maintains electrical neutrality by allowing ions to migrate and balance the movement of electrons in the external circuit.
- The electrolyte must be chemically compatible with the electrode materials to prevent unwanted reactions or precipitation (e.g., sodium sulfate is often used with copper sulfate to avoid forming insoluble salts).
5. External Circuit
- Serves as the pathway for electron flow from the anode to the cathode.
- In a Galvanic Cell: Electrons flow spontaneously, generating electrical energy as a result of a spontaneous redox reaction.
- In an Electrolytic Cell: An external power source is required to push electrons and drive a non-spontaneous redox reaction.
6. Salt Bridge:
- A U-shaped tube or porous barrier filled with an inert electrolyte such as KNO³ or Na²SO&sub4;.
- Function 1: Completes the internal circuit by allowing ion flow, preventing charge buildup in the half-cells.
- Function 2: Prevents direct mixing of oxidizing and reducing agents, ensuring that electron transfer occurs only through the external circuit.
- Function 3: Acts like a semipermeable membrane, permitting the movement of electrolyte ions while minimizing the crossover of redox-active species.
- This maintains electrical neutrality, sustains the redox reaction, and ensures continuous current flow.
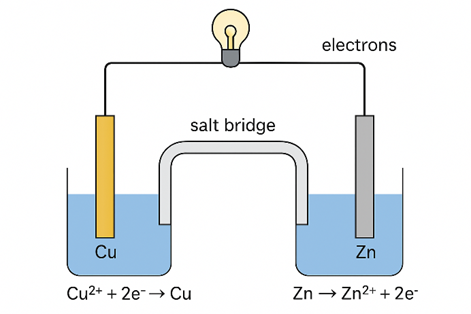
Explanation of Daniell Cell
A typical galvanic cell is designed to utilize the spontaneous redox reaction between zinc and cupric ions to generate an electric current. The setup includes a copper vessel filled with a saturated solution of copper(II) sulfate (CuSO₄), which acts as the depolarizer, and dilute sulfuric acid (H₂SO₄), serving as the electrolyte. An amalgamated zinc rod is immersed in a zinc sulfate (ZnSO₄) solution. Just below the upper edge of the copper vessel, a transparent layer holds CuSO₄ crystals in contact with the CuSO₄ solution, ensuring that the solution remains saturated throughout the operation of the cell.
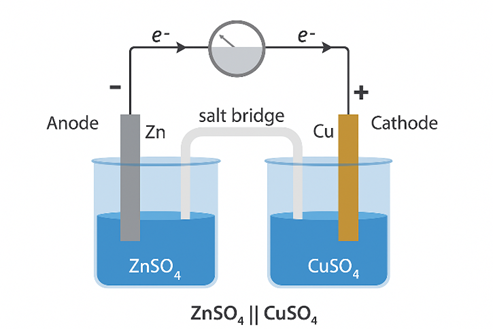
Steps Involved in Daniell Cell
- The Daniell cell can be conventionally represented as:
Zn(s) + Cu2+(aq) → Zn2+(aq) + Cu(s)
- In a Daniell cell, electrons flow from the zinc electrode to the copper electrode through an external circuit, while metal ions transition from one half-cell to the other via the salt bridge structure.
- Here, current flows from the copper electrode to the zinc electrode (that is, from cathode to anode) via the external electrical circuit interface.
Proudly Powered By


Leave a Comment