Schrödinger Wave Equation
The Schrödinger Equation is the fundamental pillar of quantum mechanics, serving as the “Newton’s Second Law” for the subatomic world. The schrodinger equation derivation can be achieved from the classical wave equation as well as from the third postulate of quantum mechanics. While classical physics uses equations to predict the exact path of an object, the Schrödinger equation describes the probability of finding a particle in a specific location.
The above mentioned second order differential equation is our popular form of Schrodinger wave equation.
What It Represents
At its core, the equation treats particles (like electrons) as wavefunctions . Instead of a hard point moving through space, the electron is viewed as a “cloud” of probability. The equation allows us to calculate how this wave evolves over time and how it is influenced by external forces (potential energy).+2
Why It’s Revolutionary
The Foundation of Chemistry: Every chemical bond and periodic table trend is ultimately a result of the solutions to this equation.
Quantization: It explains why electrons in an atom can only exist at specific, “quantized” energy levels rather than anywhere they want.
Wave-Particle Duality: It mathematically bridges the gap between a particle’s mass and its wave-like behavior.
Derivation based on Classical Wave equation
The Schrodinger wave equation can be derived from the classical wave equation as well as from the third postulate of quantum mechanics. Now though the two routes may appear completely different, the final result is just the same indicating the objectivity of the quantum mechanical system. After the failure of the Bohr atomic model to comply with the Heisenberg’s uncertainty principle and dual character proposed by Louis de Broglie in 1924, an Austrian physicist Erwin Schrodinger developed his legendary equation by making the use of wave-particle duality and classical wave equation.
The Schrödinger equation is the cornerstone of quantum mechanics. While it is often treated as a postulate, it can be conceptually derived by merging the Classical Wave Equation with the De Broglie Hypothesis regarding wave-particle duality. This bridge explains why microscopic particles like electrons behave as “matter waves.”
Schrödinger Wave Equation-Complete Lecture in Urdu/Hindi
In order to understand the concept involved, consider a wave traveling in a string along the x-axis with velocity v.
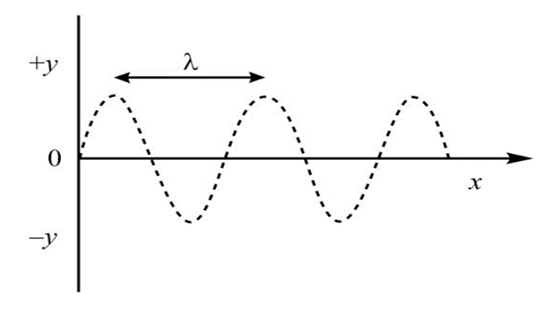
Step 1: The Classical Foundation
In classical physics, a wave (such as a vibration on a string) traveling along the x-axis is described by a second-order differential equation.It can be clearly seen that the amplitude of the wave at any time t is the function of displacement x, and the equation for wave motion can be formulated as given below.
Therefore, we can say that y is a function of x as well at t. Here, y represents the amplitude at any position x and time t, while v is the phase velocity of the wave.
Step 2: Separation of Variables (Standing Waves)
For a stationary or “standing” wave (like an electron bound in an atom), we can separate the variables. We assume the total wave function is a product of a space-dependent function f(x) and a time-dependent function f'(t):
Where f(x) and f'(t) are the functions of coordinate x and time, respectively.
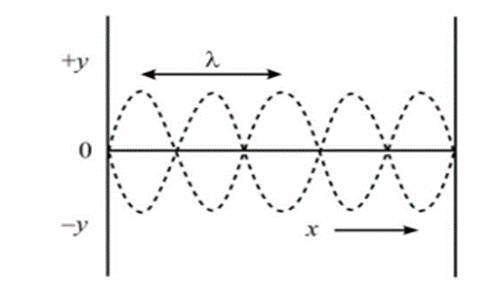
The nature of the function f(x) can be understood by taking the example of stationary or the standing wave.
A standing wave is created in a string fixed between two points with a wave traveling in one direction, and when it strikes the other end, it gets reflected with the same velocity but in negative amplitude.
This would create vibrations in that string with or without nodes depending upon the frequency incorporated. We can create fundamental mode (0 node), first overtone (1 node) or second overtone (2 nodes) just by changing the vibrational frequency. The nature of these standing or stationary waves can be understood more clearly by the diagram given below.
The is For harmonic motion, the mathematical description for such a wave motion is expressed as:
The is For harmonic motion, the mathematical description for such a wave motion is expressed as:
Where A is constant representing maximum amplitude and v is the frequency of the vibrations.
Now putting the value of 𝑓′ (𝑡) from equation (15) in equation (14), we get
Step 3: Differentiating the Time Component
To substitute this back into our classical equation, we differentiate y with respect to t twice:
Differentiating the above equation w.r.t. t we are left with
Differentiating again
Step 4: Eliminating Time Now differentiating equation (14) w.r.t x only , we get
Differentiating again
Now put the value of equation (7) and (9) in equation (1), we get
The equation (11) is now time-independent, and therefore , shows the amplitude dependence. Only upon the coordinate x. Since c=v𝞴 , (v = c/𝞴 ), yhe velocity of wave can also be replaced. By the multiplication of frequency and wavelength i.e. v= v𝞴.
The symbol of 𝑓(𝑥) is replaced by popular 𝟁(t) or simply 𝟁
Step 5: Injecting Quantum Mechanics (De Broglie)
To transform this into a quantum mechanical equation, we apply the De Broglie relation. De Broglie proposed that any particle with mass m and velocity v has a wavelength 𝞴 = h / mv , the equation becomes
Step 6: Incorporating Total Energy
In any system, Total Energy (E) is the sum of Kinetic Energy (K) and Potential Energy (V):
E = K + V
We can say that
After putting the value of from equation (18) in equation (16), we get
For three dimensional i.e. (𝑥,𝑦,𝑧), 𝑡ℎ𝑒 𝑎𝑏𝑜𝑣𝑒 𝑒𝑞𝑢𝑎𝑡𝑖𝑜𝑛 𝑐𝑎𝑛 𝑏𝑒 𝑒𝑥𝑡𝑒𝑛𝑑𝑒𝑑 𝑡𝑜 𝑓𝑜𝑙𝑙𝑜𝑤𝑖𝑛𝑔
The above mentioned second order differential equation i.e. equation (18) is our popular form of Schrodinger wave equation.
Download Complete Notes Below
Proudly Powered By


Leave a Comment